Research Articles
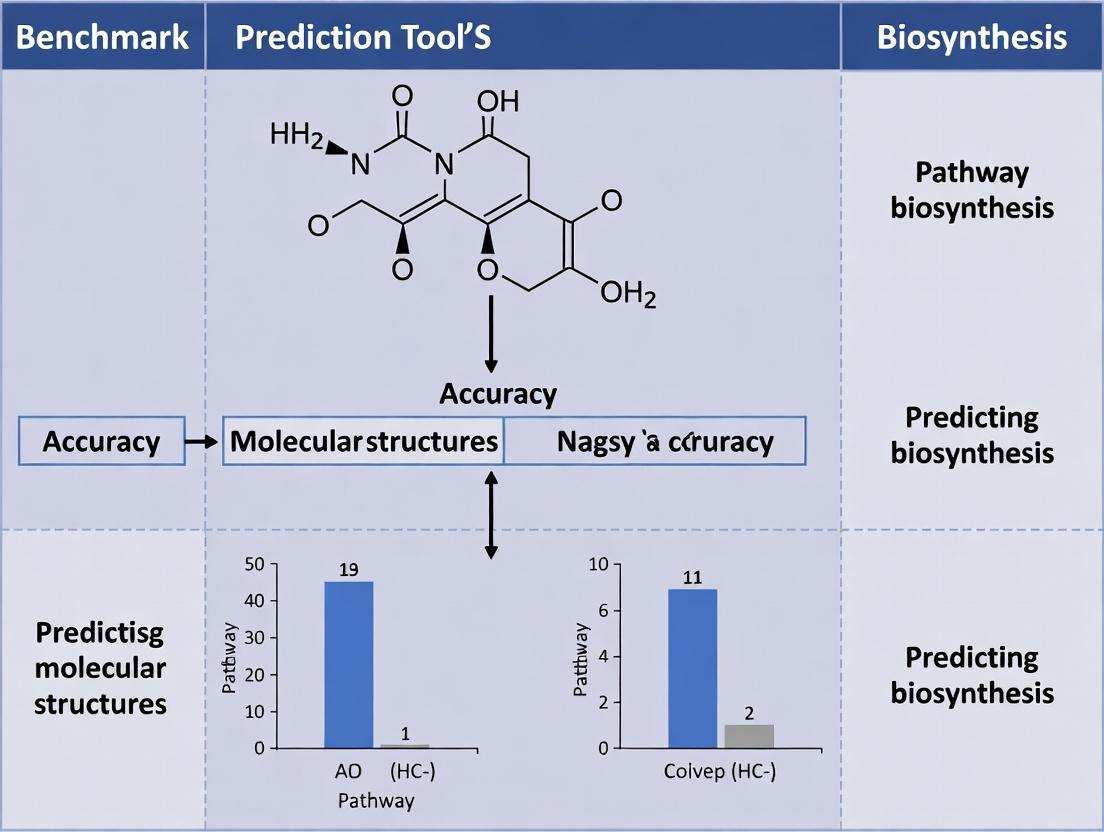
Benchmarking Bioinformatic Pathway Tools: A Practical Guide to Accuracy, Validation, and Application in Biomedical Research
This article provides a comprehensive guide for researchers and drug development professionals on evaluating the accuracy of pathway prediction tools.
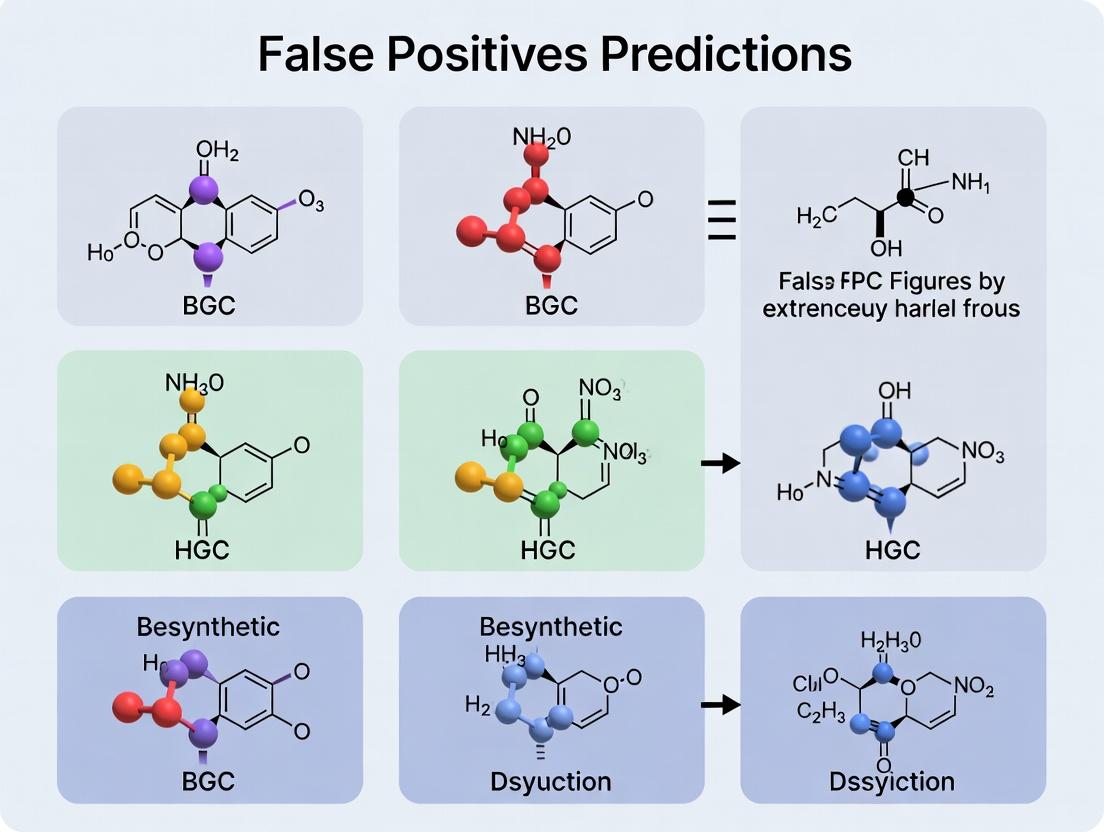
BGC Prediction False Positives: Strategies for Accuracy in Natural Product Discovery
This comprehensive article addresses the critical challenge of false positives in Biosynthetic Gene Cluster (BGC) prediction, a major bottleneck in natural product discovery pipelines.
Overcoming Expression Hurdles: A Guide to Enzyme Solubility and Post-Translational Modification in Heterologous Systems
This article provides a comprehensive guide for researchers and bioprocess scientists addressing the critical challenges of expressing functional enzymes in heterologous hosts.
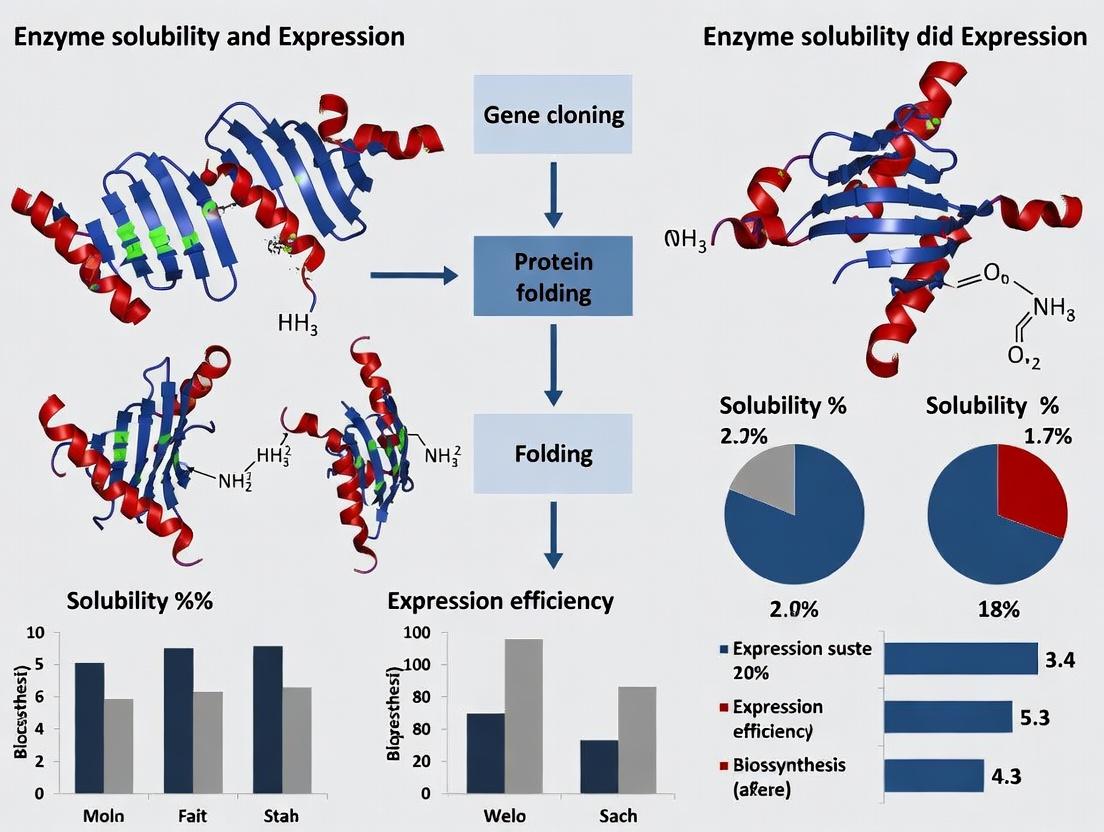
Strategies for Overcoming Enzyme Solubility and Expression Challenges in Biopharmaceutical Research
This article provides a comprehensive guide for researchers and drug development professionals facing enzyme solubility and expression issues.
Taming Enzyme Promiscuity: Advanced Strategies to Minimize Unwanted Side Products in Drug Synthesis and Biocatalysis
This article provides a comprehensive guide for researchers, scientists, and drug development professionals on addressing enzyme promiscuity and its generation of unwanted side products.
Harnessing Enzyme Promiscuity: Strategies for Novel Natural Product Discovery in Drug Development
This article provides a comprehensive overview of strategies to address and exploit enzyme promiscuity in diversity-oriented biosynthesis, aimed at accelerating drug discovery.
Beyond the Sugar Coat: Strategies to Overcome Donor Cost Challenges in Chemo-Enzymatic Glycoengineering
Chemo-enzymatic glycoengineering enables precise glycosylation for next-generation biologics and glycoconjugate vaccines, but the high cost of nucleotide sugar donors remains a critical barrier to industrial adoption.
From Noise to Knowledge: A Practical Framework for Diagnosing and Resolving Data Quality Issues in Materials Science Training Datasets
This article provides researchers, scientists, and drug development professionals with a comprehensive, actionable guide to managing data quality in materials training datasets.
Beyond the Majority: Advanced Strategies for Tackling Data Imbalance in Materials Foundation Models
This article provides a comprehensive guide for researchers, scientists, and development professionals on addressing the pervasive challenge of data imbalance in materials science foundation models.
Mastering Cofactor Balancing: The Key to Efficient Synthetic Biology and Biomanufacturing
This comprehensive guide for researchers, scientists, and drug development professionals explores the critical challenge of cofactor balancing in synthetic metabolic pathways.