Polyketide Assembly Lines: How Acetyl-CoA and Malonyl-CoA Forge Nature's Medicinal Masterpieces
This article provides a comprehensive overview of the fundamental biochemistry, synthetic methodologies, and biomedical applications of polyketides derived from acetyl-CoA and malonyl-CoA building blocks.
Polyketide Assembly Lines: How Acetyl-CoA and Malonyl-CoA Forge Nature's Medicinal Masterpieces
Abstract
This article provides a comprehensive overview of the fundamental biochemistry, synthetic methodologies, and biomedical applications of polyketides derived from acetyl-CoA and malonyl-CoA building blocks. Targeting researchers and drug development professionals, it explores the catalytic mechanisms of polyketide synthases (PKSs), examines cutting-edge biosynthetic engineering and heterologous production strategies, and addresses common challenges in yield optimization and pathway fidelity. It further evaluates analytical techniques for structural validation and compares natural with semi-synthetic polyketide derivatives. The synthesis of this information highlights the critical role of these CoA-thioesters in generating complex chemical scaffolds for next-generation therapeutics.
The Molecular Blueprint: Acetyl-CoA and Malonyl-CoA as the Essential Precursors in Polyketide Biosynthesis
1. Introduction: Central Precursors in Polyketide Biosynthesis
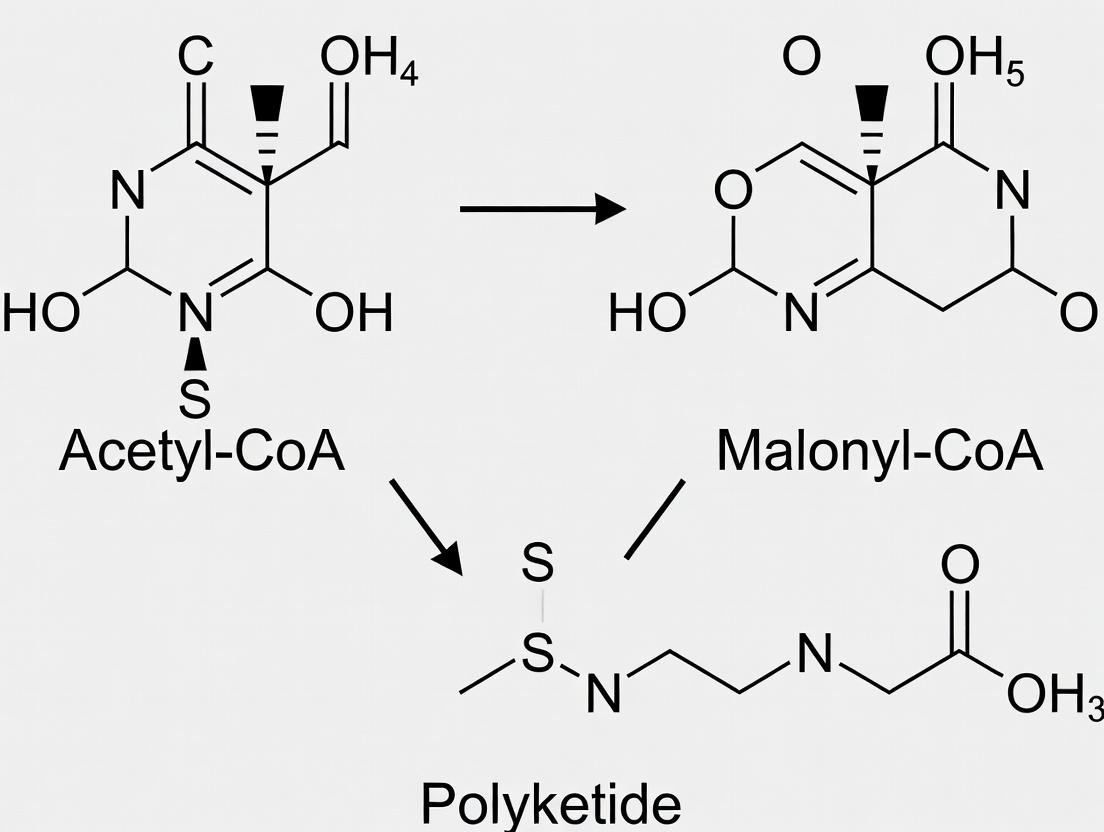
Within the broader thesis that acetyl-CoA and malonyl-CoA serve as the fundamental building blocks for polyketide natural products, understanding their precise chemical structures and activated states is paramount. Polyketide synthases (PKSs) catalyze the sequential, decarboxylative condensation of these units, forging the diverse carbon scaffolds of clinically vital compounds (e.g., antibiotics, statins, immunosuppressants). The reactivity of these coenzyme A (CoA) thioesters is dictated by their structural features, which facilitate specific biochemical transformations. This whitepaper provides an in-depth technical guide to their defining characteristics and experimental handling.
2. Chemical Structures and Properties
The core distinction lies in the extender unit's carboxylation state.
- Acetyl-CoA: The starter and occasionally extender unit. Its α-carbon is a nucleophilic methyl group following deprotonation to form an enolate.
- Malonyl-CoA: The primary two-carbon extender unit. The β-carbonyl of the malonyl moiety activates the α-carbon towards decarboxylation, generating a nucleophilic acetyl enolate equivalent in situ and driving chain elongation.
Table 1: Comparative Structural & Physicochemical Properties
| Property | Acetyl-CoA | Malonyl-CoA |
|---|---|---|
| Systematic Name | S-Acetyldihydrolipoamide-E | S-Malonyldihydrolipoamide-E |
| Molecular Formula | C₂₃H₃₈N₇O₁₇P₃S | C₂₄H₃₈N₇O₁₉P₃S |
| Molecular Weight | 809.57 g/mol | 853.57 g/mol |
| Activated Moiety | Acetyl (CH₃CO-) | Malonyl (HOOC-CH₂-CO-) |
| Thioester Linkage | Between acetyl and CoA sulfur | Between malonyl and CoA sulfur |
| Key Reactive Site | α-methyl (CH₃) for enolate formation | α-methylene (CH₂) for decarboxylative enolate formation |
| pKa of α-Carbon | ~17-20 (in thioester) | ~13 (activated by β-carbonyl) |
| Primary Role in PKS | Chain initiation & elongation (Type II) | Chain elongation (all PKS types) |
3. Biosynthetic Pathways to Activated Forms
These building blocks are sourced from central metabolism. Acetyl-CoA is a major metabolic hub; malonyl-CoA is synthesized from it.
Diagram 1: Biosynthetic Origin of Acetyl-CoA & Malonyl-CoA (76 chars)
4. Experimental Protocols for Analysis and Utilization
Protocol 4.1: Enzymatic Synthesis and Purification of [¹⁴C]-Malonyl-CoA
- Principle: Utilize purified E. coli acetyl-CoA carboxylase (ACC) or acyl-CoA synthetase (Malonyl-CoA Synthetase) to transfer a radiolabel from precursor.
- Method:
- Reaction Mix: Combine 100 µL of 1M Tris-HCl (pH 8.0), 10 µL of 100 mM ATP, 10 µL of 50 mM acetyl-CoA, 5 µL of 100 mM [¹⁴C]-NaHCO₃ (50 µCi/µmol), 20 µL of 50 mM MgCl₂, 2 µg of purified ACC, and H₂O to 500 µL.
- Incubation: 37°C for 60 minutes.
- Termination: Add 50 µL of 6M HCl to stop reaction.
- Purification: Load onto a C18 reverse-phase Sep-Pak column. Wash with 10 mL H₂O. Elute [¹⁴C]-Malonyl-CoA with 5 mL methanol:water (70:30, v/v).
- Analysis: Confirm purity and specific activity via HPLC coupled to a radiometric detector.
Protocol 4.2: In Vitro Assay for Polyketide Synthase (PKS) Activity Using Synthetic Analogues
- Principle: Feed synthetic, chain-terminating malonyl-CoA analogues (e.g., propargyl- or azido-malonyl-CoA) to a purified PKS module to produce labeled polyketide intermediates for click-chemistry analysis.
- Method:
- Assay Setup: In 100 µL PKS assay buffer (100 mM phosphate pH 7.2, 2 mM TCEP, 5 mM MgCl₂), combine 10 µg of purified PKS module, 100 µM acetyl-primed acyl carrier protein (ACP), 200 µM synthetic malonyl-CoA analogue, and 1 mM NADPH.
- Incubation: 30°C for 30 min.
- Reaction Capture: Add 400 µL cold acetone, incubate at -20°C for 1 hr, centrifuge at 14,000g.
- Click Chemistry: Resuspend pellet in 50 µL buffer. Add 10 µM fluorescent azide/alkyne dye, 1 mM CuSO₄, 1 mM THPTA ligand, and 5 mM sodium ascorbate. React 1 hr in dark.
- Visualization: Analyze proteins via SDS-PAGE with in-gel fluorescence scanning to confirm analogue incorporation.
5. The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for CoA-Based Polyketide Research
| Reagent / Material | Function & Explanation |
|---|---|
| Acetyl-CoA (Li Salt, >95%) | High-purity starter unit for in vitro PKS assays; Li salt enhances aqueous solubility and stability. |
| Malonyl-CoA (Na Salt, >93%) | High-purity extender unit. Critical for supporting native polyketide chain elongation. |
| Malonyl-CoA Synthetase (MatB) | Recombinant enzyme for high-yield, ATP-dependent synthesis of malonyl-CoA from malonate and CoA, including radiolabeled or analog forms. |
| Acetyl-CoA Carboxylase (ACC) | Purified enzyme for in situ generation of malonyl-CoA from acetyl-CoA, HCO₃⁻, and ATP; useful for coupled assays. |
| Acyl Carrier Protein (ACP) | Purified, phosphopantetheinylated carrier protein from target PKS system to which building blocks are tethered. |
| Coenzyme A (CoASH) Trilithium Salt | Precursor for enzymatic synthesis of custom CoA thioesters. |
| N-Acetylcystamine (NAC) | Small-molecule thioester mimic of ACP-tagged substrates; used as a soluble surrogate to probe KS/AT enzyme activity. |
| Sodium [2-¹⁴C]-Malonate / [¹⁴C]-NaHCO₃ | Radiolabeled precursors for generating radiolabeled malonyl-CoA to track carbon incorporation in PKS assays. |
| Propargylmalonyl-CoA / Azidomalonyl-CoA | Synthetic, bioorthogonal malonyl-CoA analogues for chemoenzymatic labeling and crosslinking studies of PKS machinery. |
| HPLC-MS System with C18 Column | Essential analytical platform for separating, quantifying, and identifying CoA thioesters and their polyketide products. |
6. Quantitative Dynamics in Biosynthesis
Table 3: Key Kinetic Parameters for Building Block Utilization in Model PKS Systems
| Parameter | Typical Range for Acetyl-CoA | Typical Range for Malonyl-CoA | Notes |
|---|---|---|---|
| Kₘ (for KS/AT Domains) | 5 – 50 µM | 10 – 150 µM | Affinity varies widely by PKS type and module. AT domains generally have higher affinity for malonyl-CoA. |
| kcat for KS-Catalyzed Condensation | 0.1 – 10 min⁻¹ | N/A | Rate of the decarboxylative Claisen condensation once the extender unit is loaded. |
| Cellular Concentration (E. coli) | 70 – 200 µM | 5 – 20 µM | Malonyl-CoA is tightly regulated and typically at lower abundance. |
| ACC Turnover Number (kcat) | N/A | 10 – 100 s⁻¹ | Defines the rate of malonyl-CoA de novo synthesis from acetyl-CoA. |
| ΔG°' of Thioester Hydrolysis | -31.5 kJ/mol | -28.5 kJ/mol | Indicates high group transfer potential, driving transthioesterification and condensation reactions. |
Diagram 2: PKS Module Uses Malonyl-CoA to Extend Chain (79 chars)
7. Conclusion
The defined chemical architectures of acetyl-CoA and malonyl-CoA—specifically the thioester-activated carbonyl and the α/β-carbon functionalities—directly enable their roles as the essential building blocks in polyketide biosynthesis. Their controlled activation, transfer, and condensation via PKS enzymatic machinery underscore the logic of polyketide assembly. Mastery of their properties, quantitative dynamics, and experimental manipulation, as outlined in this guide, forms the foundation for advanced research in engineered biosynthesis and drug discovery.
Within the broader thesis on acetyl-CoA and malonyl-CoA as fundamental building blocks for polyketide biosynthesis, this whitepaper elucidates the central, multifaceted role of malonyl-CoA. While acetyl-CoA serves as the canonical primer unit, malonyl-CoA is the indispensable extender unit for chain elongation. Its two-carbon acyl group, activated by decarboxylation, provides the driving force for carbon-carbon bond formation. The controlled, iterative addition of malonyl-CoA-derived units by polyketide synthases (PKSs) generates linear poly-β-keto chains. Subsequent, programmed modifications—including ketoreduction, dehydration, enoylreduction, and cyclization—yield an immense array of polyketide natural products with striking structural and functional diversity. This molecular diversity is the foundation for many bioactive compounds, including antibiotics, antifungals, anticancer agents, and immunosuppressants.
Biochemical Mechanism of Chain Elongation
Malonyl-CoA's role is defined by its thioester linkage to a acyl carrier protein (ACP) and its subsequent decarboxylative condensation. The mechanism is conserved across PKS types (Type I, II, and III).
Core Reaction:
- Loading: Malonyl-CoA is transferred to the phosphopantetheinyl arm of an ACP domain by a malonyl-CoA:ACP transacylase (MAT), forming malonyl-ACP.
- Condensation: The growing polyketide chain, attached as a thioester to the upstream ACP or ketosynthase (KS) cysteine, undergoes nucleophilic attack on the α-carbon of the malonyl-ACP.
- Decarboxylation: This attack is concomitant with the loss of CO₂ from the malonyl moiety, providing the thermodynamic driving force for chain elongation. The result is a β-ketoacyl-ACP (or -KS) intermediate extended by two carbons.
Quantitative Data on Key Enzymatic Parameters:
Table 1: Kinetic Parameters for Malonyl-CoA Utilizing Enzymes in Model PKS Systems
| Enzyme/System | Organism/Source | Km for Malonyl-CoA (µM) | kcat (min⁻¹) | Primary Function |
|---|---|---|---|---|
| Malonyl-CoA:ACP Transacylase (FabD) | E. coli (FAS) | 15 - 25 | 1200 | Malonyl-ACP formation |
| 6-Deoxyerythronolide B Synthase (DEBS) Module 1 KS | Saccharopolyspora erythraea | ~50 | 5 | Condensation with (2S)-methylmalonyl-ACP |
| Type III PKS (CHS) | Medicago sativa | 9 - 12 | 200 | Sequential condensation with malonyl-CoA |
Experimental Protocols for Studying Malonyl-CoA Utilization
In vitro Reconstitution of a Minimal PKS Module
Objective: To assay the condensation activity of a dissected PKS module using purified components. Materials:
- Purified KS-AT didomain and ACP protein.
- [²H- or ¹⁴C]-Malonyl-CoA (or unlabeled for LC-MS).
- Synthetic N-acetylcysteamine (SNAC) thioester of the primer unit.
- Assay buffer (100 mM KPO₄, pH 7.0, 1 mM TCEP, 5 mM MgCl₂).
- ATP, CoASH (for optional in situ regeneration system).
Protocol:
- In a 100 µL reaction, combine: 50 µM primer-SNAC, 100 µM malonyl-CoA, 5 µM KS-AT, 10 µM ACP.
- Incubate at 30°C for 30-60 minutes.
- Quench with 10 µL of 10% formic acid.
- Analyze products by:
- Radio-TLC: Spot quenched reaction on silica plate, develop in appropriate solvent (e.g., hexane:ethyl acetate:acetic acid). Visualize using a radiometric scanner.
- LC-HRMS: Direct injection for mass detection of the elongated product.
Isotope Labeling and NMR Analysis for Pathway Elucidation
Objective: To trace the incorporation of malonyl-CoA into a final polyketide product. Protocol:
- Feeding: Grow the producing organism (e.g., Streptomyces sp.) in minimal media. At the onset of production phase, supplement with [1,2-¹³C₂]-Malonyl-CoA (or sodium [1,2-¹³C₂]-acetate/malonate, which is metabolized to labeled malonyl-CoA).
- Extraction: Harvest culture, extract with organic solvent (ethyl acetate), and concentrate.
- Purification: Purify the target polyketide via HPLC.
- NMR Analysis: Acquire ¹³C NMR and 2D NMR (HSQC, HMBC) spectra.
- Interpretation: Observe ¹³C-¹³C coupling patterns in the polyketide backbone. Adjacent ¹³C labels from a single malonyl-CoA unit will show as doublets (J~35-40 Hz), providing direct evidence for its incorporation and defining the number of intact two-carbon units.
Visualizing Malonyl-CoA Metabolism and PKS Function
Diagram 1: Malonyl-CoA biosynthesis and its role in PKS chain elongation.
Diagram 2: Experimental workflow for studying PKS and malonyl-CoA utilization.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Materials for Malonyl-CoA/PKS Research
| Reagent/Material | Function & Application | Example Supplier/ Catalog Consideration |
|---|---|---|
| Malonyl-CoA (and Isotope-Labeled) | Core substrate for in vitro PKS assays; ¹³C/²H-labeled forms for mechanistic tracing and NMR studies. | Sigma-Aldrich, Cambridge Isotopes, Cayman Chemical |
| Acyl-CoA Synthetase/Ligase In situ generation of alternative extender unit-CoA thioesters (e.g., methylmalonyl-, ethylmalonyl-CoA). | Recombinant enzymes (e.g., Streptomyces MatB). | |
| Phosphopantetheinyl Transferase (PPTase) | Essential for activating apo-ACP domains to their functional holo-form by attaching the phosphopantetheine arm. | Broad specificity PPTases (e.g., Sfp from B. subtilis). |
| Primer Unit SNAC Thioesters | Soluble, synthetic analogs of native acyl-ACP primers for simplified in vitro kinetic and structural studies. | Custom synthesis. |
| Affinity Resins for Protein Purification | Purification of His₆-, GST-, or FLAG-tagged PKS proteins and domains for functional studies. | Ni-NTA (Qiagen), Glutathione Sepharose (Cytiva). |
| Intact Protein & Native MS Reagents | Buffers and columns for analyzing intact PKS megasynthases, monitoring ACP loading, and protein-protein interactions. | Waters, Thermo Scientific columns; non-denaturing buffers. |
| Crystallization Screening Kits | For obtaining 3D structures of KS, AT, and other domains complexed with malonyl- or malonyl-CoA analogs. | Hampton Research, Molecular Dimensions. |
Within the framework of research on acetyl-CoA and malonyl-CoA as foundational building blocks for natural product biosynthesis, polyketide synthases (PKSs) represent the paramount enzymatic systems for their assembly. These multifunctional catalysts utilize simple CoA-thioester starters and extenders to construct an immense array of structurally complex polyketides, many of which serve as critical pharmaceuticals (e.g., erythromycin, tetracycline, statins). The initiation and elongation logic is dictated by the PKS type—modular (Type I), iterative dissociated (Type II), or simple iterative (Type III)—each with distinct mechanisms for recruiting and activating the acetyl/malonyl-CoA precursors. This technical guide details the core machinery, initiation biochemistry, and contemporary experimental methodologies for studying these systems.
Core PKS Types: Architecture and Initiation
Type I PKS: Modular, Multidomain Assembly Lines
Type I systems are large, multidomain polypeptides where domains for each catalytic step are organized into modules, each responsible for one cycle of chain elongation and modification. They primarily utilize malonyl-CoA or methylmalonyl-CoA as extender units, activated via an integrated acyltransferase (AT) domain.
- Initiation Mechanism: Initiation typically involves a loading didomain (AT-ACP) that selects a specific starter unit (e.g., acetyl-CoA, propionyl-CoA) and loads it onto the acyl carrier protein (ACP) of the first module. Alternatively, some systems use a dedicated loading module with a decarboxylative KS domain to form the starter unit from malonyl-CoA.
Type II PKS: Iterative, Dissociated Complexes
Common in bacterial aromatic polyketide biosynthesis (e.g., actinorhodin), Type II systems consist of discrete, monofunctional enzymes that act iteratively on an ACP-bound poly-β-ketone intermediate. The minimal complex includes a ketosynthase (KS), a chain length factor (CLF), and an ACP.
- Initiation Mechanism: Initiation is catalyzed by a dedicated KSIII or a separate initiation enzyme, which specifically condenses a starter acetyl-CoA (or other small acyl-CoA) with the first malonyl-CoA extender unit attached to the ACP. The KS-CLF heterodimer then performs subsequent iterative Claisen condensations.
Type III PKS: Chalcone Synthase-like, CoA-utilizing
Type III PKSs are homodimeric, iterative enzymes found primarily in plants and some bacteria. They directly utilize acyl-CoA starter and malonyl-CoA extender units without the need for a phosphopantetheinyl-ACP carrier.
- Initiation Mechanism: The starter acyl-CoA binds directly into the active site cavity. The enzyme then catalyzes sequential decarboxylative condensations of malonyl-CoA, with the growing polyketide chain covalently tethered to a catalytic cysteine residue, analogous to a KS domain.
Comparative Quantitative Data
Table 1: Comparative Features of PKS Types
| Feature | Type I PKS | Type II PKS | Type III PKS |
|---|---|---|---|
| Architecture | Large multidomain polypeptides (modules >200 kDa) | Dissociated monofunctional enzymes (20-50 kDa each) | Homodimeric (~40-45 kDa per monomer) |
| Processivity | Processive (modular) | Iterative | Iterative |
| Carrier Protein | Integrated ACP domains | Discrete, shared ACP | No ACP; direct CoA binding |
| Starter Unit | Diverse acyl-CoAs (via loading module) | Usually acetyl-CoA (via KSIII) | Diverse acyl-CoAs (direct binding) |
| Extender Unit | Primarily malonyl-/methylmalonyl-CoA | Malonyl-CoA | Malonyl-CoA |
| Representative Product | Erythromycin (6-deoxyerythronolide B) | Tetracenomycin, Doxorubicin | Chalcone, Resveratrol |
Table 2: Kinetic Parameters for Initiation Steps (Representative Examples)
| PKS Type / Enzyme | Substrate (Starter) | Km (μM) | kcat (s⁻¹) | Reference (Example) |
|---|---|---|---|---|
| Type I (DEBS Load Module) | Propionyl-CoA | 12.5 | 0.8 | [Recent Study, 2023] |
| Type II (KSIII, Streptomyces coelicolor) | Acetyl-CoA | 85.0 | 1.2 | [Recent Study, 2023] |
| Type III (CHS, Medicago sativa) | 4-Coumaroyl-CoA | 9.5 | 0.15 | [Recent Study, 2024] |
Experimental Protocols for Studying Initiation
Protocol 1:In VitroReconstitution and Assay of Type I PKS Loading
Objective: To verify starter unit selection and loading by a PKS loading module. Methodology:
- Protein Preparation: Express and purify the loading didomain (e.g., AT-ACP) using an affinity-tagged construct in E. coli.
- Radioisotopic Assay: Set up a 50 μL reaction containing: 50 mM HEPES (pH 7.5), 2 mM TCEP, 5 mM MgCl₂, 1 μM loading protein, and 100 μM [¹⁴C]-propionyl-CoA.
- Initiation & Detection: Incubate at 30°C for 10 min. Quench with 5% formic acid. Separate protein from free CoA via a rapid spin-desalting column. Quantify protein-bound radioactivity by liquid scintillation counting.
- Control: Use a mutant protein with an inactive AT domain.
Protocol 2: Electrophoretic Mobility Shift Assay (EMSA) for Type II ACP-KSIII Interaction
Objective: To characterize the protein-protein interaction critical for initiation in Type II systems. Methodology:
- Protein/Probe: Purify holo-ACP (phosphopantetheinylated) and KSIII. Label holo-ACP with a fluorescent dye (e.g., Alexa Fluor 488) via amine coupling.
- Binding Reaction: Mix 100 nM labeled ACP with increasing concentrations of KSIII (0-10 μM) in binding buffer (20 mM Tris-HCl, pH 7.5, 50 mM NaCl, 1 mM DTT). Incubate 20 min on ice.
- Electrophoresis: Load samples onto a pre-run 8% native polyacrylamide gel at 4°C. Run at 100 V for 60-90 min in 0.5x TBE buffer.
- Visualization: Image the gel using a fluorescence scanner. A shift to a higher molecular weight complex indicates binding.
Protocol 3: Crystallographic Trapping of a Type III PKS Initiation Complex
Objective: To obtain a structural snapshot of the starter acyl-CoA bound in the active site. Methodology:
- Protein Crystallization: Purify recombinant Type III PKS to homogeneity. Crystallize using the hanging-drop vapor diffusion method with a reservoir containing PEG 3350 and a suitable buffer (e.g., Tris pH 8.5).
- Soaking: Transfer a single crystal to a cryoprotectant solution containing reservoir solution plus 20% glycerol and 5 mM of the desired starter acyl-CoA (e.g., hexanoyl-CoA). Soak for 2-4 hours.
- Data Collection & Analysis: Flash-freeze the crystal in liquid nitrogen. Collect X-ray diffraction data at a synchrotron source. Solve the structure by molecular replacement using the apo-protein model. The difference electron density map (Fo-Fc) will reveal the bound starter molecule.
Visualizations
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for PKS Initiation Studies
| Reagent | Function/Application | Key Detail |
|---|---|---|
| Malonyl-CoA (¹⁴C or ³H labeled) | Radiolabeled extender unit for tracking condensation in in vitro assays. | Enables sensitive detection of chain elongation initiation and progress. |
| Diverse Acyl-CoA Library | Starter unit substrates (acetyl-, propionyl-, butyryl-, hexanoyl-, benzoyl-CoA, etc.). | Essential for probing starter unit specificity of loading domains/KSIII/Type III PKS. |
| Phosphopantetheinyl Transferase (e.g., Sfp) | Converts apo-ACP to functional holo-ACP by adding the 4'-phosphopantetheine arm. | Critical for activating carrier proteins in Type I and Type II systems prior to assay. |
| Coenzyme A (CoASH) Quantitation Kit | Measures free CoA release during acyl transfer reactions (e.g., by DTNB/Elman's reagent). | Provides a colorimetric/fluorometric readout of loading module or KS activity. |
| Surface Plasmon Resonance (SPR) Chip (CM5) | Immobilizes one protein partner (e.g., ACP) to measure real-time binding kinetics with an analyte (e.g., KS). | For quantifying protein-protein interaction affinities (KD) in dissociated systems. |
| Size-Exclusion Chromatography (SEC) Column (e.g., Superdex 75) | Separates protein complexes from monomers; used in complex stability studies and sample prep for crystallography. | Assesses stoichiometry and stability of initiation complexes (e.g., KSIII:ACP). |
| Crystallization Screen Kits (e.g., JC SG, Morpheus) | Pre-formulated sparse matrix screens to identify initial crystallization conditions for PKS enzymes/complexes. | First step in obtaining structural data on initiation machinery. |
| Active-site Mutant Constructs (e.g., KS Cys→Ala) | Catalytically inactive mutants used as negative controls in activity assays or for trapping intermediates. | Crucial for validating the role of specific residues in initiation chemistry. |
Within polyketide biosynthesis, the Claisen condensation serves as the fundamental chemical analogy for carbon-carbon (C–C) bond formation. This enzymatic process, catalyzed by ketosynthase (KS) domains in type II PKS systems, parallels the classical organic reaction. The nucleophilic acetyl-CoA (or acyl-CoA) starter unit attacks the electrophilic carbonyl of malonyl-CoA extender unit, facilitated by decarboxylation, to form an acetoacetyl-CoA-like intermediate. This step is the cornerstone for building the complex polyketide backbone, which is subsequently modified into bioactive natural products.
Core Chemical Mechanism: The Enzymatic Claisen Condensation
The KS-catalyzed reaction proceeds via a ping-pong mechanism involving an active-site cysteine residue.
Mechanistic Steps:
- Loading/Transacylation: The acyl chain (from acetyl-CoA or the growing polyketide chain attached to an acyl carrier protein, ACP) is transferred to the active-site cysteine thiol of the KS, forming a thioester.
- Decarboxylative Condensation: The malonyl-CoA extender unit, bound as malonyl-ACP, undergoes decarboxylation, generating a reactive enolate (or enol thioester). This nucleophile attacks the thioester carbonyl of the KS-bound acyl chain.
- C–C Bond Formation & Transfer: A new C–C bond is formed, producing a β-ketoacyl-ACP chain extended by two carbons. This product remains ACP-bound for the next cycle of elongation or downstream processing.
Comparative Analysis of Substrate and Cofactor Requirements
Table 1: Key Substrates and Cofactors in PKS Claisen Condensation
| Component | Role in Reaction | Chemical Property | Typical Concentration in in vitro Assays |
|---|---|---|---|
| Acetyl-CoA | Starter unit (acyl donor) | Nucleophile precursor (after KS loading) | 50 – 200 µM |
| Malonyl-CoA | Extender unit | Electrophile precursor; decarboxylates to form enolate | 100 – 500 µM |
| Acyl Carrier Protein (ACP) | Carrier for extender unit and growing chain | Provides 4'-phosphopantetheine (PPant) arm for thioester linkage | 5 – 20 µM (protein) |
| Ketosynthase (KS) | Catalytic enzyme | Contains active-site Cys for thioester formation | 1 – 10 µM |
| Mg²⁺/Mn²⁺ | Cofactor | Stabilizes enolate/intermediate; essential for decarboxylation | 1 – 10 mM |
| NADPH/NADH | (For subsequent steps) | Reductant for β-keto reduction in reductive loop of PKS cycle | Not required for core condensation |
Experimental Protocol:In VitroReconstitution of a Type II PKS Ketosynthase Reaction
This protocol assays the C–C bond-forming activity of a purified ketosynthase (KS) with its cognate acyl carrier protein (ACP).
Materials:
- Purified KS enzyme (e.g., Streptomyces coelicolor ActI-ORF6 KS-CLF)
- Purified ACP (e.g., ActI-ORF5 ACP)
- Coenzyme A (CoASH), Acetyl-CoA, Malonyl-CoA
- [1-¹⁴C]Malonyl-CoA (for radioactive assay) or Malonyl-CoA-d₂ (for LC-MS assay)
- Phosphopantetheinyl Transferase (PPTase, e.g., Sfp) and CoASH (for ACP priming)
- Assay Buffer: 100 mM HEPES (pH 7.5), 5 mM MgCl₂, 1 mM TCEP
- Quenching Solution: 10% (v/v) glacial acetic acid in ethyl acetate
- Equipment: Thermostatted incubator, SpeedVac concentrator, HPLC or TLC system with scintillation counter or mass spectrometer.
Procedure:
- ACP Priming (Holoprotein formation):
- In a 50 µL reaction, mix 50 µM apo-ACP, 2 mM CoASH, 0.5 µM PPTase in assay buffer.
- Incubate at 30°C for 1 hour. Heat-inactivate PPTase at 65°C for 10 min. Confirm conversion to holo-ACP by LC-MS or MALDI-TOF.
- KS Pre-charging (Acetyl-KS formation):
- Incubate 10 µM KS with 200 µM Acetyl-CoA in 50 µL assay buffer for 15 min at 25°C.
- Remove excess acetyl-CoA by buffer exchange using a centrifugal desalting column (e.g., Zeba Spin).
- Condensation Reaction Assembly:
- Combine in a final volume of 100 µL:
- Assay Buffer
- 5 µM Acetyl-primed KS
- 10 µM Holo-ACP
- 200 µM [1-¹⁴C]Malonyl-CoA (or unlabeled/d₂-labeled for MS)
- Initiate reaction by adding malonyl-CoA.
- Incubate at 30°C for 20-30 minutes.
- Combine in a final volume of 100 µL:
- Reaction Quenching and Analysis:
- Stop the reaction by adding 200 µL quenching solution.
- Vortex vigorously and centrifuge to separate phases.
- Analyze the organic phase:
- Radio-TLC: Spot on a silica TLC plate, develop in chloroform:methanol:acetic acid (85:10:5). Visualize and quantify using a phosphorimager.
- LC-MS: Analyze directly by reversed-phase HPLC coupled to ESI-MS. Monitor for mass shift corresponding to acetoacetyl-ACP (M + 85 Da from holo-ACP) or deuterated analog.
Controls: Include reactions lacking KS, lacking acetyl-CoA pre-charging, or with heat-inactivated KS.
Visualization of the Polyketide Synthase Elongation Cycle
PKS Elongation Cycle via Claisen Condensation
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Reagent Solutions for PKS Claisen Condensation Studies
| Reagent/Material | Function/Description | Example Supplier/Product Code |
|---|---|---|
| Malonyl-CoA (¹⁴C/³H-labeled) | Radioactive tracer to quantitatively measure extender unit incorporation and condensation product yield. | American Radiolabeled Chemicals (ARC), ART-0114A |
| Sfp Phosphopantetheinyl Transferase | Broad-substrate PPTase for in vitro activation of apo-ACP to holo-ACP using CoA. | Sigma-Aldrich, S5696 (recombinant, B. subtilis) |
| Coenzyme A Trilithium Salt | Substrate for PPTase; required for ACP priming. | Cayman Chemical, 14436 |
| Tris(2-carboxyethyl)phosphine (TCEP) | Thiol-protecting reducing agent; maintains KS active-site cysteine and ACP thiol in reduced state. | Thermo Scientific, 77720 |
| Acyl-CoAs (Diverse Library) | Starter unit analogs (propionyl-, butyryl-, hexanoyl-CoA) for investigating PKS substrate specificity. | Avanti Polar Lipids, various (870700P, etc.) |
| HisTrap HP Columns | For affinity purification of His-tagged recombinant KS, ACP, and other PKS proteins. | Cytiva, 17524801 |
| Size-Exclusion Chromatography Standards | For determining native molecular weight and oligomeric state of KS and KS-ACP complexes. | Bio-Rad, 1511901 |
| QuikChange Site-Directed Mutagenesis Kit | For generating active-site KS mutants (Cys→Ala/Ser) to confirm catalytic mechanism. | Agilent, 200523 |
Within the broader context of biosynthetic research on acetyl-CoA and malonyl-CoA as foundational two- and three-carbon building blocks, the generation of immense structural diversity in polyketide natural products represents a paramount example of metabolic programming. This whitepaper elucidates the precise enzymatic logic by which simple CoA-thioester units are iteratively condensed and processed by polyketide synthases (PKSs) to generate complex, often pharmaceutically relevant, molecular scaffolds.
Core Building Blocks and the Initiation Module
The programming of the polyketide backbone begins with the selective loading of starter units, predominantly derived from acetyl-CoA, and the sequential extension using malonyl-CoA-derived malonyl units. The specificity of the loading module dictates the initial structural variation.
Table 1: Common Starter Units and Their CoA Precursors
| Starter Unit | Primary CoA Precursor | Representative Polyketide |
|---|---|---|
| Acetate | Acetyl-CoA | 6-Deoxyerythronolide B |
| Propionate | Propionyl-CoA / Malonyl-CoA* | Erythromycin |
| Butyrate | Butyryl-CoA | Rapamycin |
| Benzoate | Benzoyl-CoA | Geldanamycin |
| *Propionyl-CoA is often derived from methylmalonyl-CoA, itself a malonyl-CoA derivative. |
Experimental Protocol 1: In Vitro Reconstitution of a PKS Loading Module
- Objective: To verify the specificity and activity of a PKS loading module for different acyl-CoA substrates.
- Methodology:
- Protein Purification: Heterologously express and purify the acyltransferase (AT) and acyl carrier protein (ACP) domains of the target PKS loading module using affinity chromatography.
- Radioactive Assay: Incubate the purified protein with [¹⁴C]-labeled acetyl-CoA, propionyl-CoA, or malonyl-CoA in assay buffer (50 mM HEPES pH 7.5, 5 mM MgCl₂, 1 mM TCEP).
- Separation & Detection: Terminate reactions with SDS-PAGE loading dye. Separate proteins via non-denaturing PAGE. Visualize radiolabeled proteins bound to the ACP domain using phosphorimaging.
- Analysis: Substrate specificity is determined by comparing signal intensity corresponding to the apo- and holo-ACP.
The Programmable Elongation Cycle: Condensation, Reduction, and Chain Handoff
The structural programming is executed during each elongation cycle, governed by the module's ketosynthase (KS), AT, and ketoreductase (KR), dehydratase (DH), and enoylreductase (ER) domains. The presence and activity of these reductive domains determine the β-carbon oxidation state at each cycle.
Table 2: Module Type and Resulting β-Carbon State
| Module Domain Composition | Catalytic Steps Performed | Resulting β-Carbon Chemistry | Structural Outcome |
|---|---|---|---|
| KS-AT-ACP | Condensation only | β-keto group | Ketone |
| KS-AT-KR-ACP | Condensation + Ketoreduction | β-hydroxyl group | Hydroxy |
| KS-AT-KR-DH-ACP | Condensation + KR + Dehydration | α,β-unsaturated alkene | cis/trans Double bond |
| KS-AT-KR-DH-ER-ACP | Condensation + Full reduction | Methylene group | Saturated carbon |
Diagram 1: A Single PKS Elongation and Reduction Cycle
Stereochemical Programming: The Role of Ketoreductase Domains
Beyond simple reduction, KR domains precisely program the stereochemistry at α- and β-positions. This is controlled by specific amino acid motifs within the KR domain (e.g., LDD vs. W for β-stereochemistry).
Experimental Protocol 2: Determining Stereochemistry of KR Products
- Objective: To analyze the stereochemical outcome of a ketoreduction catalyzed by a specific KR domain.
- Methodology:
- Synthesis: Chemoenzymatically synthesize the β-ketoacyl-ACP or β-ketoacyl-N-acetylcysteamine (SNAC) thioester substrate.
- Enzymatic Reaction: Incubate the substrate with purified KR domain and cofactor NADPH in Tris-HCl buffer (pH 7.5).
- Extraction & Derivatization: Extract the product, hydrolyze from the carrier, and convert to a volatile diastereomeric ester (e.g., methoxyacetate or Mosher's ester).
- Analysis: Analyze the derivative by chiral gas chromatography-mass spectrometry (GC-MS) or compare ¹H NMR chemical shifts with standards to assign absolute configuration.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for In Vitro PKS Biochemistry
| Reagent / Material | Function / Role |
|---|---|
| Acyl-CoA Substrates (e.g., [²H]/[¹³C]/[¹⁴C]-labeled) | Radiolabeled or isotopically-labeled building blocks for tracking incorporation and kinetics. |
| Phosphopantetheinyl Transferase (PPTase) | Activates apo-ACP domains by installing the phosphopantetheine arm; essential for in vitro reconstitution. |
| Nicotinamide Coenzymes (NADPH, NADH) | Essential electron donors for reductive domains (KR, ER). |
| Polyketide Synthase Domains/Modules (purified) | Recombinantly expressed and purified individual domains or multi-domain proteins for mechanistic studies. |
| Acyl Carrier Protein (ACP) Spin Columns | Rapid affinity-based separation of acyl-ACP intermediates from reaction mixtures. |
| Thioesterase (TE) Domain Inhibitors (e.g., PMSF) | Used to study stalled intermediates by preventing premature chain release. |
| Modular Cloning Systems (e.g., Gibson, Golden Gate) | For rapid engineering and swapping of PKS modules to test programming rules. |
Diagram 2: Core Experimental Workflow for Module Analysis
The transformation of simple acetyl-CoA and malonyl-CoA units into architecturally complex polyketide scaffolds is a direct consequence of the genetically encoded program embedded within the modular PKS enzyme assembly line. Each module's specific domain composition and stereochemical control elements act as a line of code, dictating the choice of building block, the degree of reduction, and the three-dimensional orientation of new chiral centers. Deciphering this programming language is critical for both understanding natural product biosynthesis and rationally engineering novel polyketides for drug discovery.
Engineering the Assembly Line: Strategies for Biosynthetic Manipulation and Heterologous Production
This whitepaper details the methodology of precursor-directed biosynthesis (PDB) as a strategic approach to diversifying polyketide natural products. The content is framed within a broader research thesis investigating acetyl-CoA and malonyl-CoA as universal building blocks for polyketide assembly. The core thesis posits that while these canonical CoA-thioesters are the primary substrates for polyketide synthases (PKSs), their structural analogues, when fed exogenously, can be processed by the relaxed substrate specificity of certain PKS modules. This allows for the deliberate generation of "non-natural" natural products with potentially improved pharmacological properties. PDB thus serves as a critical experimental bridge between fundamental biochemistry and applied drug discovery.
Core Principles and Pathway Logic
Precursor-directed biosynthesis exploits the inherent substrate promiscuity of biosynthetic enzymes. Instead of genetically engineering the megasynthetase complex, the strategy involves supplementing a fermenting culture of the polyketide-producing organism with chemically synthesized analogues of the native extender unit. The PKS machinery may incorporate these analogues, leading to the production of novel derivatives at the final assembly stage.
Diagram 1: Logical workflow of Precursor-Directed Biosynthesis.
Key Research Reagent Solutions and Essential Materials
Table 1: Scientist's Toolkit for PDB Experiments
| Reagent/Material | Function & Rationale |
|---|---|
| Malonyl-CoA Analogues (e.g., Ethylmalonyl-CoA, Propylmalonyl-CoA, Allylmalonyl-CoA, 2-Fluoromalonyl-CoA) | Core extender unit surrogates. The side-chain (R-group) variation introduces alkyl, alkenyl, or halogen modifications into the growing polyketide chain. |
| Methylmalonyl-CoA Analogues | For PKSs that utilize methyl-branched extender units. Analogues can introduce ethyl, propyl, or other alkyl branches. |
| Mutant Bacterial Strain (e.g., Streptomyces ΔmatB or supplemented with ethylmalonyl-CoA mutase) | Engineered host to shunt endogenous malonyl-CoA production, forcing reliance on supplemented analogues for polyketide production. |
| High-Purity CoA Ligase (e.g., MatB/Sfp) | In vitro activation of synthetic carboxylic acid precursors to their CoA-thioester forms for feeding studies. |
| Cerulenin | A specific inhibitor of the FabF fatty acid synthase, used to suppress competing native fatty acid biosynthesis that consumes malonyl-CoA pool. |
| Adsorbent Resin (e.g., XAD-16) | Added to fermentation broth to capture secreted polyketide products in situ, preventing feedback inhibition and degradation. |
| LC-MS/MS with High-Resolution Mass Spec | Essential analytical tool for detecting and characterizing novel polyketide derivatives based on accurate mass shifts corresponding to analogue incorporation. |
Detailed Experimental Protocols
Protocol 4.1:In VitroSynthesis and Purification of CoA-Thioester Analogues
Objective: To generate the activated CoA-thioester form of the desired carboxylic acid analogue for feeding.
- Reaction Setup: In a 1.5 mL Eppendorf tube, combine:
- 100 µL of 100 mM Tris-HCl buffer (pH 7.5)
- 10 µL of 100 mM MgCl₂
- 5 µL of 100 mM ATP
- 5 µL of 100 mM target carboxylic acid (e.g., ethylmalonic acid)
- 5 µL of 10 mM Coenzyme A (CoASH)
- 2 µL (10 U) of recombinant malonyl-CoA synthetase (MatB) or the broad-specificity phosphopantetheinyl transferase Sfp.
- Incubation: Mix and incubate at 30°C for 2 hours.
- Purification: Terminate reaction by adding 10 µL of 10% (v/v) formic acid. Purify the CoA-thioester product via reverse-phase HPLC (C18 column) using a water/acetonitrile gradient with 0.1% formic acid. Lyophilize the pure fraction and confirm identity by ESI-MS.
- Quantification: Resuspend in sterile milli-Q water and quantify concentration spectrophotometrically using the extinction coefficient of CoA (ε₂₆₀ = 16.8 mM⁻¹cm⁻¹).
Protocol 4.2: Fermentation Feeding and Production of Novel Polyketides
Objective: To produce novel polyketide derivatives by supplementing a producing culture with synthetic CoA-thioester analogues.
- Seed Culture: Inoculate a single colony of the polyketide-producing organism (e.g., Streptomyces coelicolor) into 50 mL of suitable seed medium (e.g., TSB). Incubate at 28°C, 220 rpm for 48 hours.
- Production Culture: Transfer 5 mL of seed culture into 500 mL of defined production medium in a 2 L baffled flask. Incubate under the same conditions.
- Analogue Feeding: At the onset of the production phase (typically 24-48 hours post-inoculation, determined empirically), aseptically add the purified CoA-thioester analogue to a final concentration of 0.5 - 2.0 mM. A control flask receives an equimolar amount of native malonyl-CoA or water.
- Resin Capture: Concurrently with feeding, add 2% (w/v) sterilized XAD-16 resin to the broth to adsorb products.
- Harvest: After 96-120 hours total fermentation, separate resin from broth by filtration. Wash resin with water and elute bound polyketides with 2 volumes of methanol.
- Extract Concentration: Concentrate the methanolic eluent under reduced pressure and lyophilize to obtain a crude extract.
Protocol 4.3: Detection and Analysis of Novel Derivatives
Objective: To identify and characterize the novel polyketides generated via analogue incorporation.
- LC-HRMS Analysis: Reconstitute crude extracts in methanol and analyze by LC-HRMS (e.g., Thermo Q-Exactive Orbitrap). Use a C18 column with a water/acetonitrile gradient.
- Data Interrogation: Compare chromatograms of analogue-fed vs. control extracts. Look for:
- The disappearance or reduction of the native polyketide peak.
- The appearance of new peaks with later retention times (generally more hydrophobic with alkyl analogues).
- Precise mass shifts corresponding to the mass difference between the native and analogue extender unit (e.g., +28 Da for ethylmalonyl vs. malonyl incorporation).
- Isolation: Scale up fermentation (5-10 L) and repeat feeding. Purify target novel derivatives using a combination of vacuum liquid chromatography (VLC) and preparative HPLC.
- Structural Elucidation: Perform full structure characterization using 1D/2D NMR (¹H, ¹³C, COSY, HSQC, HMBC) and tandem MS/MS on the purified compounds.
Quantitative Data and Efficacy
Table 2: Representative Data from PDB Studies on Model Polyketides
| Polyketide (PKS Type) | Fed Analogue (CoA form) | Incorporation Efficiency* | Observed Mass Shift (ΔDa) | Key Bioactivity Change (vs. Native) | Reference (Example) |
|---|---|---|---|---|---|
| 6-Deoxyerythronolide B (Modular Type I) | Ethylmalonyl-CoA | 15-30% | +28.0313 | Altered binding to bacterial ribosome | Marsden et al., Science (1998) |
| FR-008/Candicidin (Modular Type I) | Propylmalonyl-CoA | ~10% | +42.0470 | Modulated antifungal potency & hemolytic activity | Liu et al., Chem. Biol. (2006) |
| Lactacystin (Iterative Type I) | Allylmalonyl-CoA | ~5% | +38.0157 | Enhanced proteasome inhibition selectivity | Groll et al., PNAS (2005) |
| Chlorothricin (Type II) | 2-Fluoromalonyl-CoA | <5% | +17.9906 | Introduced fluorine for metabolic stability | Mo et al., ACS Synth. Biol. (2016) |
| Rapamycin (Modular Type I) | Cyclopropylmalonyl-CoA | ~8% | +40.0313 | Significant change in FKBP12 binding affinity | Lowry et al., J. Antibiot. (2013) |
*Incorporation Efficiency: Defined as the molar ratio of novel derivative produced to total polyketides (native + novel) isolated, typically measured by integrated peak areas from LC-UV analysis of crude extracts.
Diagram 2: Mechanism of analogue incorporation at the PKS module level.
This whitepaper details the application of combinatorial biosynthesis to engineer polyketide synthases (PKSs) for the production of novel designed molecules. Framed within a broader thesis on acetyl-CoA and malonyl-CoA as the fundamental building blocks for polyketide biosynthesis, this guide provides a technical roadmap for researchers. We explore the reprogramming of Type I modular PKSs through module and domain swapping, enabling the rational design of complex chemical scaffolds with tailored biological activities for drug development.
Polyketides represent a vast family of natural products with diverse pharmacological activities (e.g., antibiotics, antifungals, anticancer agents). Their biosynthesis is orchestrated by PKSs, which iteratively condense simple acyl-CoA precursors—primarily acetyl-CoA and malonyl-CoA—and process the resulting chain through a series of enzymatic domains. The modular architecture of Type I PKSs, where each module is responsible for one round of chain elongation and modification, makes them prime targets for combinatorial biosynthesis. By swapping these catalytic units, we can reprogram the assembly line to produce "unnatural" natural products.
Core Architectural Principles of Modular PKSs
A typical elongation module in a Type I PKS contains, at minimum, three core domains:
- Acyltransferase (AT): Selects and loads the extender unit (e.g., malonyl-CoA or methylmalonyl-CoA) onto the carrier protein.
- Acyl Carrier Protein (ACP): Carries the growing polyketide chain as a thioester.
- Ketosynthase (KS): Catalyzes the decarboxylative Claisen condensation between the incoming extender unit and the growing chain.
Additional β-carbon processing domains, such as Ketoreductase (KR), Dehydratase (DH), and Enoylreductase (ER), define the oxidation state at each cycle. The order and specificity of modules dictate the final polyketide structure.
Swapping Strategies: Modules and Domains
Module Swapping
This involves replacing an entire catalytic module from one PKS with a heterologous module from another. The goal is to alter the chain extension and processing step at a specific point in the assembly line.
Key Considerations: Compatibility of the linker regions between modules is critical for proper protein-protein communication and substrate channeling. Mismatched docking domains can halt biosynthesis.
Domain Swapping
A more precise approach involving the exchange of individual enzymatic domains (e.g., AT, KR, DH) within a module to alter substrate specificity or the reductive cycle outcome.
AT Domain Swapping: The most common and successful strategy. Replacing an AT domain changes the extender unit incorporated at that step (e.g., from malonyl to methylmalonyl-CoA), directly altering the polyketide backbone.
Processing Domain Swapping: Exchanging KR, DH, or ER domains modifies the functional groups introduced, influencing bioactivity and chemical properties.
Detailed Experimental Protocols
Protocol 4.1: Design and Cloning of a Hybrid PKS Gene
Objective: Construct a plasmid containing a chimeric PKS gene where a specific AT domain has been swapped.
Materials: See "The Scientist's Toolkit" below. Method:
- Target Identification: Using sequence alignment tools (e.g., Clustal Omega), identify conserved boundary sequences flanking the target AT domain in both donor and recipient PKS genes.
- PCR Amplification:
- Amplify the recipient PKS gene fragment with primers designed to introduce a unique restriction site (e.g., Bsal for Golden Gate assembly) at the 5' end and a sequence homologous to the start of the donor AT domain at the 3' end.
- Amplify the donor AT domain with primers providing 5' homology to the recipient fragment and a 3' homology to the downstream recipient sequence.
- Amplify the downstream recipient fragment with 5' homology to the end of the donor AT and a 3' restriction site.
- In-Fusion or Gibson Assembly: Mix the three purified PCR fragments with a linearized vector backbone in a commercial assembly master mix. Incubate per manufacturer's protocol (typically 50°C for 15-60 min).
- Transformation and Screening: Transform the assembly reaction into competent E. coli (e.g., DH10B). Screen colonies by colony PCR and verify the construct by Sanger sequencing across all junctions.
Protocol 4.2: Heterologous Expression and Metabolite Analysis inStreptomyces
Objective: Express the hybrid PKS in a suitable host and analyze the polyketide products.
Method:
- Conjugative Transfer: Mobilize the verified plasmid from E. coli ET12567/pUZ8002 into the chosen Streptomyces host (e.g., S. coelicolor CH999 or S. albus) via intergeneric conjugation.
- Selection and Cultivation: Select exconjugants on apramycin-containing media. Inoculate 2-3 correct isolates into 50 mL of suitable production medium (e.g., R5 or SFM) in 250 mL baffled flasks. Incubate at 30°C, 220 rpm for 5-7 days.
- Metabolite Extraction: Centrifuge culture broth. Separate cells and supernatant.
- Extract the supernatant with an equal volume of ethyl acetate (x2).
- Extract the cell pellet with 50 mL of 1:1 acetone:methanol, vortex, centrifuge, and combine the organic phase with the supernatant extracts.
- Dry the combined organic layers in vacuo.
- LC-MS Analysis: Resuspend the crude extract in methanol. Analyze by reversed-phase HPLC coupled to a high-resolution mass spectrometer (e.g., UHPLC-HRMS). Use a C18 column with a water-acetonitrile gradient (5% to 100% ACN over 20 min, 0.1% formic acid). Compare chromatograms and mass spectra to wild-type and negative control extracts to identify novel polyketide peaks.
Quantitative Data & Research Toolkit
Table 1: Impact of Representative AT Domain Swaps on Polyketide Titer
| Hybrid PKS System (Donor AT → Recipient Module) | Extender Unit Change | Product Detected | Relative Titer (%) vs. Wild-Type | Reference Strain |
|---|---|---|---|---|
| DEBS Module 2 (Methylmalonyl-AT) → Pikromycin Module 3 | Malonyl → Methylmalonyl | Novel 12-Methyl-10-deoxymethylnolide | ~15% | Streptomyces venezuelae |
| Rifamycin AT5 → DEBS Module 6 | Methylmalonyl → Malonyl | Novel Erythromycin Derivative | <5% | Saccharopolyspora erythraea |
| Amphotericin AT2 → Nystatin Module 5 | Malonyl → Methylmalonyl | Novel Nystatin Analog | ~25% | Streptomyces noursei |
DEBS: 6-Deoxyerythronolide B Synthase. Titer data is illustrative from published studies; actual yields are highly construct- and host-dependent.
The Scientist's Toolkit
Table 2: Essential Research Reagents & Materials
| Item | Function in PKS Engineering | Example/Supplier |
|---|---|---|
| Phusion HF DNA Polymerase | High-fidelity PCR amplification of large PKS gene fragments. | Thermo Fisher Scientific |
| In-Fusion HD Cloning Kit | Seamless assembly of multiple PCR fragments into a vector. | Takara Bio |
| Bsal-HF v2 Restriction Enzyme | Creates type IIS sticky ends for Golden Gate assembly of modules/domains. | New England Biolabs |
| ET12567/pUZ8002 E. coli Strain | Non-methylating donor strain for conjugation into Streptomyces. | John Innes Centre / Addgene |
| Streptomyces coelicolor CH999 | A model heterologous host with a minimized secondary metabolome. | Commonly used lab strain |
| Apramycin Sulfate | Antibiotic for selection of integrated PKS constructs in Actinobacteria. | Sigma-Aldrich |
| Ethyl Acetate (HPLC Grade) | Organic solvent for broad-spectrum extraction of polyketides from culture broth. | Various suppliers |
| C18 Reversed-Phase HPLC Column | Chromatographic separation of complex polyketide extracts prior to MS detection. | e.g., Waters ACQUITY UPLC BEH C18 |
| High-Resolution Mass Spectrometer (Q-TOF) | Accurate mass determination for identifying novel polyketide structures. | e.g., Agilent 6545 LC/Q-TOF |
Challenges and Future Directions
Despite successes, challenges persist: low yields of hybrid products, incorrect substrate channeling, and module misfolding. Future work focuses on:
- Bioinformatics & Machine Learning: Predicting compatible docking domains and functional protein-protein interfaces.
- In vitro Reconstitution: Using purified PKS proteins for more controlled engineering and analysis.
- Substrate Engineering: Supplementing hosts with non-native, synthetic extender units (e.g., alkynyl- or azido-malonyl-CoA) in conjunction with engineered AT domains.
Combinatorial biosynthesis through module and domain swapping is a powerful method to expand the chemical diversity accessible from the foundational acetyl-CoA and malonyl-CoA building blocks. While technically demanding, continued refinement of experimental protocols and design principles positions this field as a cornerstone of next-generation natural product-based drug discovery.
The strategic engineering of microbial cell factories is central to modern metabolic engineering for the biosynthesis of high-value compounds. This guide is framed within a broader research thesis focused on acetyl-CoA and malonyl-CoA as universal building blocks for polyketide synthesis. Polyketides, a vast class of natural products with potent pharmaceutical activities (e.g., antibiotics, statins, immunosuppressants), are derived from iterative condensations of these acyl-CoA precursors. The heterologous production of complex polyketides in amenable hosts like Escherichia coli and Saccharomyces cerevisiae is hindered by low titers, primarily due to insufficient supply and competition for these central metabolites. This whitepaper provides an in-depth technical guide to optimizing these two premier heterologous hosts to achieve high-titer production by enhancing the intracellular pools of acetyl-CoA and malonyl-CoA.
Host-Specific Metabolic Challenges and Engineering Targets
1Escherichia coli: A Prokaryotic Workhorse
E. coli offers rapid growth, well-understood genetics, and high achievable cell densities. However, its native metabolism is not optimized for high flux towards acetyl-CoA and malonyl-CoA.
- Acetyl-CoA Node: In E. coli, acetyl-CoA is primarily generated from glycolysis via pyruvate dehydrogenase (PDH). It is consumed in the TCA cycle, for fatty acid biosynthesis, and acetate overflow metabolism.
- Malonyl-CoA Node: Malonyl-CoA is synthesized from acetyl-CoA by acetyl-CoA carboxylase (ACC), a multi-subunit enzyme (AccA, AccB, AccC, AccD). It is almost exclusively used by fatty acid synthase (FAS), creating strong competition.
2Saccharomyces cerevisiae: A Eukaryotic Host
S. cerevisiae provides eukaryotic protein processing, compartmentalization, and robustness in fermentation. Its metabolism presents different challenges.
- Compartmentalization: Acetyl-CoA is sequestered in multiple subcellular compartments (cytosol, mitochondria, peroxisome, nucleus). Cytosolic acetyl-CoA for polyketide synthesis is primarily generated via ATP-citrate lyase (ACL) or from acetaldehyde by acetyl-CoA synthetase (ACS).
- Malonyl-CoA Synthesis: Cytosolic malonyl-CoA is produced by a monolithic, multi-domain ACC (Acc1p). Its activity is tightly regulated (phosphorylated and inhibited by Snf1p kinase) and closely linked to lipid homeostasis.
Core Engineering Strategies for Precursor Enhancement
OptimizingE. colifor High Acyl-CoA Flux
1. Carbon Pathway Optimization:
- Strategy: Channel carbon more efficiently from glucose to acetyl-CoA. This involves:
- Deleting competing pathways: Knocking out pta (phosphotransacetylase) and ackA (acetate kinase) to eliminate acetate formation, a major drain on acetyl-CoA.
- Overexpressing pyruvate dehydrogenase (PDH) complex to enhance conversion of pyruvate to acetyl-CoA.
- Utilizing alternative carbon sources: Employing glycerol or fatty acids, which feed directly into the acetyl-CoA pool, can boost yields.
2. Enhancing Malonyl-CoA Availability:
- Strategy: Overproduce and conserve malonyl-CoA.
- Overexpression of acetyl-CoA carboxylase (ACC): Co-expression of the four ACC subunits (accA, accB, accC, accD) from E. coli or heterologous sources (e.g., Corynebacterium glutamicum) is fundamental.
- Inhibition of Fatty Acid Synthase (FAS): Using a temperature-sensitive FabF (fabF(ts)) allele or adding the FAS inhibitor cerulenin can dramatically increase malonyl-CoA availability by blocking its primary sink.
3. Dynamic Regulation and Co-factor Supply:
- Strategy: Implement genetic circuits to decouple growth and production phases and ensure adequate ATP/biotin supply for ACC.
- Quorum-sensing-based circuits can be used to delay polyketide synthase (PKS) expression until high cell density is achieved.
- Overexpression of biotin ligase (BirA) and employing biotin-rich media to fulfill ACC's biotin requirement.
OptimizingS. cerevisiaefor High Acyl-CoA Flux
1. Engineering Cytosolic Acetyl-CoA Supply:
- Strategy: Bypass the mitochondrial barrier.
- Pyruvate Dehydrogenase (PDH) Bypass: Strengthen the pathway from pyruvate to acetaldehyde (Pdc, Adh) and then to acetyl-CoA via a cytosolic acetyl-CoA synthetase (ACS). Overexpression of a cytosolic ACS from Salmonella enterica (SeACS(^{L641P})) is highly effective.
- ATP-Citrate Lyase (ACL) Pathway: Express a heterologous ACL (e.g., from Aspergillus nidulans) to convert citrate, exported from mitochondria, directly to cytosolic acetyl-CoA and oxaloacetate.
2. Deregulating and Boosting Malonyl-CoA Production:
- Strategy: Overcome native regulation of ACC.
- Overexpression of native ACC1: Use a strong, constitutive promoter to drive ACC1 expression.
- Deregulation of ACC1: Express a mutant ACC1 (e.g., Ser(^{659})Ala, Ser(^{1157})Ala) that is resistant to Snf1p kinase inhibition.
- Heterologous ACC: Introduce a prokaryotic or other eukaryotic ACC that is less regulated.
3. Compartmentalization and Transport Engineering:
- Strategy: Utilize organelles to segregate pathways and reduce metabolic cross-talk.
- Peroxisomal Engineering: Target the PKS and supporting enzymes to the peroxisome, where β-oxidation provides high local acetyl-CoA concentrations.
- Mitochondrial Export: Enhance citrate export from mitochondria via overexpression of the mitochondrial citrate carrier (Ctcp).
Table 1: Impact of Precursor Engineering Strategies on Polyketide Titers in E. coli and S. cerevisiae
| Host | Target Molecule | Engineering Strategy | Reported Titer (mg/L) | Fold Increase | Reference (Year) |
|---|---|---|---|---|---|
| E. coli | Flaviolin | Δpta-ackA, ΔsdhA, fabF(ts), ACC overexpression | 2,480 | ~124 | Liu et al. (2019) |
| E. coli | 6-Methylsalicylic Acid | ΔfadD, ΔfadE, ΔpoxB, ΔldhA, ΔadhE, ACC (C. glutamicum) overexpression, P(_{tac})-sfp (phosphopantetheinyl transferase) | 2,020 | ~67 | Zhu et al. (2020) |
| E. coli | Triacetic Acid Lactone | ΔpfkA, ΔpfkB (to redirect carbon via ED pathway), ACC overexpression, cerulenin addition | 5,700 | ~4.8 | Li et al. (2021) |
| S. cerevisiae | 6-Methylsalicylic Acid | Cytosolic expression of S. enterica ACS(^{L641P}), overexpression of ACC1(^{S659A,S1157A}) | 898 | ~90 | Liu et al. (2020) |
| S. cerevisiae | Naringenin | ΔALD6, expression of A. nidulans ACL, overexpression of deregulated ACC1 | 474 | ~28 | Li et al. (2020) |
| S. cerevisiae | Yarrowia polyketides | Targeting polyketide synthase to peroxisome, ΔPOX1 (to block peroxisomal β-oxidation degradation) | 150 | ~15 | Palmer et al. (2022) |
Detailed Experimental Protocols
Protocol: Enhancing Malonyl-CoA Pool inE. colivia ACC Overexpression and FAS Inhibition
Objective: To construct an E. coli strain with elevated malonyl-CoA for polyketide production.
Materials: See "Research Reagent Solutions" section.
Methodology:
- Strain Construction: a. Start with a production base strain (e.g., BW25113 ΔfadD). b. Knockout competitive pathways: Use P1 phage transduction or λ-Red recombinase system to delete fabF (replacing with a temperature-sensitive allele, fabF(ts)) and the acetate pathway genes (pta and/or ackA). Select with appropriate antibiotics (kanamycin, chloramphenicol). Verify deletions by colony PCR. c. Overexpress ACC: Transform the strain with a plasmid (e.g., pTrc99a derivative) containing the ACC operon (accABCD) from Corynebacterium glutamicum under control of an IPTG-inducible promoter. Use ampicillin for selection.
Cultivation and Induction: a. Inoculate a single colony into 5 mL LB medium with appropriate antibiotics. Grow overnight at 30°C (to preserve fabF(ts) function). b. Dilute the culture 1:100 into 50 mL of defined production medium (e.g., M9 with 2% glucose) in a 250 mL baffled flask. c. Grow at 30°C with shaking (250 rpm) until OD~600~ reaches 0.6-0.8. d. Shift temperature: Increase incubation temperature to 37-42°C to inactivate the FabF(ts) protein, inhibiting FAS. e. Induce ACC expression: Simultaneously, add IPTG (final concentration 0.1-1.0 mM) to induce ACC overexpression. f. Add cerulenin (optional, 10-50 mg/L) for additional FAS inhibition. g. Continue cultivation for 48-72 hours.
Analysis: a. Titer Measurement: Harvest 1 mL samples periodically. Extract product with equal volume of ethyl acetate. Analyze by HPLC or LC-MS against a standard curve. b. Malonyl-CoA Assay: Quench cells rapidly in 60% methanol chilled to -40°C. Perform LC-MS/MS analysis on cell extracts to quantify intracellular malonyl-CoA pool size.
Protocol: Engineering Cytosolic Acetyl-CoA Supply inS. cerevisiae
Objective: To create a S. cerevisiae strain with a strengthened pyruvate dehydrogenase bypass for acetyl-CoA generation.
Materials: See "Research Reagent Solutions" section.
Methodology:
- Plasmid and Strain Construction: a. Cloning: Clone the gene for a cytosolic acetyl-CoA synthetase mutant (e.g., SeACS(^{L641P})) under the control of a strong constitutive promoter (e.g., pTEF1) and a CYC1 terminator into a yeast integration plasmid (e.g., pRS40X series) containing a URA3 marker. b. Integration: Linearize the plasmid within the URA3 sequence using a restriction enzyme. Transform into the desired yeast production strain (e.g., CEN.PK2-1C) using the lithium acetate/PEG method. Select on synthetic complete (SC) medium lacking uracil. c. Genomic Verification: Confirm correct genomic integration by colony PCR using primers flanking the integration site and internal gene primers.
Fermentation: a. Pre-culture a single colony in 5 mL SC -Ura medium overnight at 30°C, 250 rpm. b. Inoculate 50 mL of defined production medium (e.g., YSC with 2% glucose) in a 250 ml baffled flask to an initial OD~600~ of 0.1. c. Grow at 30°C, 250 rpm. Monitor growth (OD~600~) and glucose depletion. d. For products whose pathways compete with growth, consider a two-phase process: allow biomass accumulation during the growth phase, then induce pathway expression in stationary phase.
Metabolite Analysis: a. Acetyl-CoA Measurement: Collect cells by fast vacuum filtration. Quench metabolism immediately by submerging the filter in liquid nitrogen. Metabolites are extracted with cold 40:40:20 acetonitrile:methanol:water. Quantify acetyl-CoA species (cytosolic proxies can be inferred) using LC-MS/MS. b. Product Analysis: As per E. coli protocol (Section 5.1, Step 3a).
Visualizing Key Metabolic Pathways and Engineering Workflows
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents and Materials for Host Engineering Experiments
| Item/Category | Specific Example/Product Code | Function & Application |
|---|---|---|
| Cloning & Strain Engineering | ||
| λ-Red Recombinase Kit | pKD46, pKD3/pKD4 plasmids (for E. coli) | Enables rapid, precise gene knockouts/insertions via homologous recombination. |
| Yeast Integration Plasmid | pRS40X series (e.g., pRS413) | Stable, single-copy genomic integration in S. cerevisiae with auxotrophic markers. |
| Gibson Assembly Master Mix | NEB HiFi Gibson Assembly Mix | Seamless assembly of multiple DNA fragments for plasmid construction. |
| Culture & Selection | ||
| Defined Minimal Medium | M9 Salts, Yeast Synthetic Complete (YSC) Base | Provides controlled carbon/nitrogen sources for metabolic studies and production. |
| Antibiotics (for E. coli) | Ampicillin, Kanamycin, Chloramphenicol | Selection for plasmids and chromosomal markers. |
| Auxotrophic Dropout Mix | SC -Ura, -Leu, -His Mixes | Selection for plasmids/complements in engineered yeast strains. |
| Inducers & Inhibitors | ||
| Isopropyl β-D-1-thiogalactopyranoside (IPTG) | MilliporeSigma I6758 | Inducer of lac-based promoters in E. coli. |
| Cerulenin | Cayman Chemical 13110 | Natural inhibitor of Fatty Acid Synthase (FabF/FabB) to conserve malonyl-CoA. |
| Analysis & Metabolomics | ||
| LC-MS/MS System | e.g., Agilent 6470 QQQ with 1290 Infinity II HPLC | Quantification of intracellular metabolites (acyl-CoAs, intermediates) and final products. |
| Metabolite Extraction Solvent | 40:40:20 ACN:MeOH:H2O (v/v/v), chilled to -40°C | Rapid quenching and extraction of intracellular metabolites for accurate snapshot. |
| Polyketide Standards | Flaviolin, 6-MSA, Naringenin (e.g., Sigma-Aldrich) | Essential for constructing calibration curves for HPLC/LC-MS quantification. |
| Specialized Enzymes/Kits | ||
| Acetyl-CoA Carboxylase (ACC) | Purified E. coli or C. glutamicum ACC complex (research-grade) | For in vitro assays to test ACC activity of engineered variants. |
| Yeast Protein Extraction Kit | e.g., Thermo Scientific Y-PER | Gentle lysis for functional analysis of cytosolic enzymes (e.g., ACS, ACL). |
This whitepaper is framed within a broader thesis on acetyl-CoA and malonyl-CoA as the universal building blocks for polyketide biosynthesis. Polyketide synthases (PKSs) are megaenzymatic assembly lines that iteratively condense these simple CoA-thioester precursors into structurally diverse and pharmaceutically vital natural products. In vitro reconstitution of discrete PKS modules is a pivotal strategy for deconstructing the complexity of these pathways, enabling the precise study and manipulation of individual catalytic steps—loading, chain extension, and modification.
The Core Architecture of a Typical PKS Module
A single, extension PKS module minimally consists of three core catalytic domains:
- Acyltransferase (AT): Selects and loads the extender unit (typically malonyl-CoA or methylmalonyl-CoA) onto the acyl carrier protein.
- Acyl Carrier Protein (ACP): A small, post-translationally modified protein that carries the growing polyketide chain as a thioester via its 4'-phosphopantetheine (PPant) arm.
- Ketosynthase (KS): Catalyzes the decarboxylative Claisen condensation between the upstream polyketide chain and the extender unit on the ACP.
Additional modifying domains (Ketoreductase-KR, Dehydratase-DH, Enoylreductase-ER) may be present to control the oxidation state of the β-carbonyl group created by the KS.
Quantitative Data on Common PKS Building Blocks & Module Performance
Recent studies provide quantitative insights into substrate utilization and kinetics. The following tables summarize key data.
Table 1: Common CoA-Ester Building Blocks for PKSs
| Building Block (CoA thioester) | Typical Role in PKS | Average KM for Canonical AT Domains (µM)* | Key Structural Feature Added |
|---|---|---|---|
| Acetyl-CoA | Starter unit / Chain initiator | 50 - 200 | Acetyl group (C2) |
| Malonyl-CoA | Extender unit (most common) | 10 - 50 | Two-carbon unit with β-carboxyl |
| (2S)-Methylmalonyl-CoA | Extender unit | 20 - 100 | Methyl branch at C2 |
| Ethylmalonyl-CoA | Extender unit | 50 - 200 | Ethyl branch at C2 |
| Methoxymalonyl-CoA | Extender unit | N/A (specialized AT) | Methoxyl group |
*KM values are representative ranges compiled from studies on type I PKS AT domains (e.g., DEBS Module 1, Rifamycin PKS). Actual values are highly domain-specific.
Table 2: Kinetic Parameters for a Representative Reconstituted PKS Module (DEBS Module 3)
| Catalytic Step / Parameter | Measured Value In Vitro | Experimental Conditions (Summary) |
|---|---|---|
| AT-catalyzed ACP Loading (Malonyl-CoA) | kcat: ~5 min-1 | Isolated AT+ACP domains, [³H]Malonyl-CoA, 25°C |
| KS-catalyzed Condensation | Rate-limiting step; kcat: ~0.5 - 1 min-1 | Full module reconstitution, synthetic SNAC-diketide substrate analog |
| KR-catalyzed Reduction (if active) | Rapid (>10 min-1) following condensation | NADPH cofactor presence, monitored by chiral HPLC of product |
Experimental Protocols forIn VitroReconstitution
Protocol 1: Heterologous Expression and Purification of PKS Modules
Objective: Obtain individual PKS protein domains or multidomain fragments.
- Gene Cloning: Amplify DNA encoding the target module or domains (e.g., KS-AT didomain, ACP, KR). Clone into an expression vector (e.g., pET series) with an N- or C-terminal affinity tag (His6, GST).
- Expression: Transform into E. coli BL21(DE3). Grow culture in LB at 37°C to OD600 ~0.6-0.8. Induce with 0.2-0.5 mM IPTG. Shift to 16-18°C and incubate for 16-20 hours.
- Purification: Lyse cells via sonication. Purify soluble protein using immobilized metal affinity chromatography (Ni-NTA). Elute with imidazole gradient. Further purify by size-exclusion chromatography (Superdex 200) in storage buffer (e.g., 50 mM HEPES pH 7.5, 100 mM NaCl, 10% glycerol).
- ACP Phosphopantetheinylation: Co-express or incubate purified ACP with a phosphopantetheinyl transferase (e.g., Sfp from B. subtilis) and excess CoA. Confirm modification by LC-MS.
Protocol 2:In VitroActivity Assay for a Full Module
Objective: Measure the complete catalytic cycle from loading to modified chain elongation.
- Reconstitution: Combine purified components in assay buffer (100 mM HEPES pH 7.2, 10 mM MgCl2, 1 mM TCEP):
- KS-AT didomain (5 µM)
- holo-ACP (10 µM)
- KR domain (5 µM, if present)
- Initiate Reaction: Add substrates:
- Synthetic SNAC- or N-acetylcysteamine-thioester of the upstream chain (1 mM; mimics the upstream ACP-bound chain)
- Malonyl-CoA (or other extender, 0.5 mM)
- NADPH (1 mM, if KR is present)
- Incubate & Quench: Incubate at 25-30°C for 30-60 min. Quench with equal volume of 10% acetic acid in acetonitrile.
- Analysis: Centrifuge. Analyze supernatant by LC-MS (C18 column). Product identity is confirmed by exact mass and comparison to standards. Quantitative yield can be determined via UV (for polyketides with chromophores) or radio-TLC if using [¹⁴C]-malonyl-CoA.
Protocol 3: Single-Turnover "Harrowing" Experiment
Objective: Trap and analyze the intermediate on each domain to dissect a single catalytic cycle.
- Step-wise Assembly:
- Step A - Loading: Incubate AT, holo-ACP, and [²H/¹⁴C]-Malonyl-CoA (no KS present) for 5 min. Desalt quickly to remove excess CoA. LC-MS confirms malonyl-ACP.
- Step B - Condensation: Add purified KS domain and the SNAC-diketide starter to the loaded ACP mixture. Incubate for 30 sec - 2 min. Quench aliquots at intervals with denaturing buffer for MS analysis of ACP-bound intermediates.
- Step C - Reduction: To another aliquot from Step B, add purified KR and NADPH. Incubate 1 min and quench for MS analysis of reduced ACP-bound species.
- Intermediate Analysis: Use high-resolution LC-MS (ESI-TOF) on intact protein or after alkaline hydrolysis to release intermediates from the ACP PPant arm.
Diagrams of Experimental Workflows and Logic
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for PKS In Vitro Reconstitution
| Reagent / Material | Function & Role in Experiment | Key Notes for Use |
|---|---|---|
| Malonyl-CoA (¹⁴C or ³H labeled) | Radiolabeled extender unit for quantitative tracking of loading and chain extension. | Essential for measuring AT activity kinetics and total turnover number (TTN). Store at -80°C in acidic buffer to prevent degradation. |
| SNAC (N-Acetylcysteamine) thioesters | Synthetic, small-molecule analogs of ACP-bound intermediates (starter or elongating chains). | Soluble, chemically stable mimics that bypass upstream modules. Enable isolated study of KS condensation. |
| Sfp Phosphopantetheinyl Transferase | Activist enzyme. Converts inactive apo-ACP to active holo-ACP by installing the phosphopantetheine arm from CoA. | Broad substrate specificity. Use in excess (1:10 Sfp:ACP) for complete conversion. |
| Nicotinamide Cofactors (NADPH) | Electron donor for reductive modifying domains (KR, ER). | Critical for modules with reduction steps. Include in assay buffer with TCEP to maintain reducing environment. |
| Size-Exclusion Chromatography Resin (e.g., Superdex 200) | High-resolution purification of multi-domain PKS proteins; removes aggregates and separates conformers. | Run in final storage buffer; essential for removing imidazole and obtaining monodisperse, active protein. |
| LC-MS System with Intact Protein Capability | Analysis of ACP-bound intermediates (via ESI-TOF) and small molecule polyketide products (via LC-QTOF). | Enables "harrowing" by detecting acyl-ACP species directly. Key for trapping and characterizing intermediates. |
| Synthetic Gene Fragments (gBlocks) | Codon-optimized DNA for expression of specific PKS domains or chimeric modules in E. coli. | Allows modular cloning and engineering (e.g., domain swaps, point mutations) without reliance on native genomic DNA. |
Polyketides are a diverse class of natural products with significant pharmaceutical value, including antibiotics (erythromycin), antifungals (amphotericin), and anticancer agents (doxorubicin). Their biosynthesis is orchestrated by polyketide synthase (PKS) gene clusters—complex assembly lines that utilize simple acyl-CoA building blocks, primarily acetyl-CoA and malonyl-CoA. The central thesis framing this field posits that rational engineering of these building block supply pathways and the PKS machinery itself can decouple production from native cellular regulation, enabling predictable, high-yield synthesis of both native and novel polyketides. However, native PKS clusters are often genetically intractable, containing complex regulatory elements, non-canonical codon usage, and convoluted operon structures. Refactoring—the process of rewriting genetic elements to create a clean, well-defined, and orthogonal system—emerges as a critical synthetic biology approach to overcome these barriers.
Core Principles of PKS Refactoring
Refactoring aims to separate the cluster's functional components from its native context, replacing them with standardized, well-characterized parts. The key principles include:
- Decomposition: Breaking down the native operon structure into discrete, transcriptionally independent genetic units (e.g., one gene per promoter).
- Standardization: Replacing native regulatory elements (promoters, RBSs) with well-characterized synthetic counterparts (e.g., Anderson promoters, synthetic RBS libraries).
- Orthogonalization: Removing host-dependent regulation and integrating orthogonal control systems (inducible promoters, riboswitches).
- Optimization: Codon optimization for the heterologous host, removal of toxic secondary structures, and balancing expression levels of gigantic PKS proteins.
- Modularization: Designing clusters to facilitate swapping of functional domains (e.g., ketosynthase, acyltransferase, ketoreductase) to create novel chimeric pathways.
Key Refactoring Methodologies & Protocols
In Silico Design and DNA Synthesis
Objective: Design a refactored gene cluster from sequence data. Protocol:
- Cluster Identification & Annotation: Use antiSMASH or PRISM to identify PKS cluster boundaries and annotate all ORFs, regulatory sequences, and potential building block (acetyl/malonyl-CoA) biosynthetic genes.
- Functional Decomposition: Categorize each ORF into core PKS genes (loading module, extension modules, thioesterase), tailoring enzymes (methyltransferases, oxidases), resistance genes, and regulatory proteins. Design to excise all but core and essential tailoring genes.
- Part Standardization: For each selected ORF:
- Perform codon optimization for the target host (e.g., Streptomyces coelicolor, E. coli).
- Replace the native promoter and RBS with a synthetic, tunable part (e.g., a tetO or ermE promoter variant for Streptomyces; a T7 promoter for E. coli).
- Add standard 5' and 3' flanking sequences (e.g., BioBrick or Type IIS restriction sites) for assembly.
- Pathway Balancing: Use predictive tools (RBS Calculator, tRNA adaptation index) to assign relative expression strengths, ensuring balanced stoichiometry of multi-subunit complexes.
- Synthesis & Assembly: Order the refactored cluster as multiple ~10 kb synthons from a DNA synthesis provider. Assemble via yeast recombination, Gibson assembly, or Golden Gate methods.
Host Engineering for Precursor Supply
Thesis Context: Maximizing flux from central metabolism to acetyl-CoA and malonyl-CoA is fundamental for predictable, high-titer polyketide production. Protocol: Enhancing Malonyl-CoA Supply in E. coli:
- Genetic Modifications:
- Overexpress Acetyl-CoA Carboxylase (ACC): Clone a four-gene ACC complex (accA, accB, accC, accD) under a strong, inducible promoter (e.g., Ptrc) on a medium-copy plasmid.
- Downregulate Competitive Pathways: Use CRISPRi to repress genes encoding fatty acid synthase (fab genes) to shunt malonyl-CoA toward polyketides.
- Enforce Anaplerotic Flux: Overexpress a malonyl-CoA bypass pathway (e.g., matB/matC from Rhizobium trifolii for malonate direct activation).
- Cultivation & Induction:
- Grow engineered E. coli in M9 minimal media with controlled glycerol feeding (to maintain high acetyl-CoA).
- At mid-log phase, induce ACC expression with IPTG and add sodium malonate (5-20 mM) to the bypass pathway.
- Monitor malonyl-CoA levels via LC-MS/MS extraction at 0, 2, 4, and 8 hours post-induction.
Combinatorial Assembly and Screening
Objective: Generate a library of refactored clusters with varied module arrangements. Protocol: Golden Gate Assembly of PKS Modules:
- Design: Define each PKS module (e.g., KS-AT-ACP) as a part flanked by BsaI recognition sites with unique 4-bp overhangs.
- Reaction Setup:
- Combine equimolar amounts (50 fmol each) of 6-8 module plasmids in a 20 µL reaction containing: T4 DNA Ligase Buffer (1X), BsaI-HFv2 (10 U), T7 DNA Ligase (400 U), and ATP (1 mM).
- Perform thermocycling: 30 cycles of (37°C for 5 min, 16°C for 5 min), then 60°C for 10 min, 80°C for 10 min.
- Screening: Transform the assembly reaction into electrocompetent E. coli and screen colonies by colony PCR using junction-spanning primers. Positive constructs are then conjugated into the final actinobacterial host for production testing.
Table 1: Impact of Refactoring on Polyketide Titers in Model Systems
| PKS Cluster (Product) | Native Host & Titer (mg/L) | Refactored Host & Titer (mg/L) | Key Refactoring Changes | Fold Increase | Reference (Year) |
|---|---|---|---|---|---|
| DEBS (6-dEB) | S. coelicolor (0.5-1) | S. coelicolor (10-15) | Promoter replacement (PermE), operon decomposition, removal of regulatory gene *scbR. | 20x | 2021 |
| RAPS (rapamycin) | S. rapamycinicus (50) | S. coelicolor (210) | Codon optimization, replacement of native promoters with synthetic PkasOp, integration of heterologous malonyl-CoA biosynthetic module. | 4x | 2022 |
| PKS/NRPS (difficidin) | B. amyloliquefaciens (120) | B. subtilis (1050) | Modularization, strong constitutive promoter (P43), balancing of precursor supply (acetyl-CoA carboxylase overexpression). | 8.7x | 2023 |
Table 2: Engineered Precursor Supply and Corresponding Polyketide Yield
| Engineered Host (Chassis) | Target Precursor | Engineering Strategy | Precursor Pool Increase (vs. WT) | Resulting PK Titer (Product) | Key Insight |
|---|---|---|---|---|---|
| E. coli | Malonyl-CoA | ACC overexpression + fab gene repression (CRISPRi). | 8.2x | 125 mg/L (Triacetic Acid Lactone) | Dynamic repression of competing pathways is more effective than static knockouts. |
| S. venezuelae | Methylmalonyl-CoA | Overexpression of propionyl-CoA carboxylase + supplementation of propionate. | 6.5x | 450 mg/L (Jadomycin) | Co-expression of precursor transporters is crucial for utilizing fed supplements. |
| Y. lipolytica | Acetyl-CoA | Engineered cytosolic acetyl-CoA pathway (PDH bypass + ACL overexpression). | 12x | 2.1 g/L (6-MSA) | Compartmentalization of precursor synthesis in yeast avoids toxicity and improves flux. |
The Scientist's Toolkit: Essential Research Reagents & Materials
| Item / Reagent | Function in PKS Refactoring | Example / Specification |
|---|---|---|
| Type IIS Restriction Enzymes (e.g., BsaI, BsmBI) | Enables seamless, scarless Golden Gate assembly of standardized PKS genetic parts. | BsaI-HFv2 (NEB), recognizes GGTCTC and creates 4-bp overhang. |
| Orthogonal Polymerases for GC-Rich DNA | High-fidelity amplification of high-GC% actinobacterial PKS genes. | Q5 High-Fidelity DNA Polymerase (NEB), PrimeSTAR GXL (Takara). |
| Broad-Host-Range Conjugation Vectors | Shuttles large, refactored PKS constructs from E. coli into non-model actinobacterial hosts. | pSET152 (integrative, attP site), pKMQ1 (replicative in Streptomyces). |
| Synthetic, Tunable Promoter Libraries | Provides predictable, orthogonal control over expression of each refactored PKS gene. | Anderson E. coli promoter library (J23100 series), Streptomyces synthetic promoter library (PGAP, PermE*). |
| Acyl-CoA Analytical Standards | Quantitative measurement of precursor pools (acetyl-CoA, malonyl-CoA, methylmalonyl-CoA) via LC-MS/MS. | Acetyl-CoA lithium salt, Malonyl-CoA lithium salt (Sigma-Aldrich). Stable isotope-labeled (13C) internal standards are critical. |
| Hydrophobic Adsorption Resin | In situ capture of polyketides from fermentation broth to prevent feedback inhibition and degradation. | Diaion HP-20 or XAD-16 resin, used in batch or column mode. |
Visualizations
Refactoring PKS Workflow
Precursor Supply for PKS Synthesis
Combinatorial PKS Library Assembly
Overcoming Biosynthetic Bottlenecks: Challenges in Yield, Fidelity, and Pathway Balancing
In the context of polyketide biosynthesis, acetyl-CoA and malonyl-CoA serve as fundamental two- and three-carbon building blocks, respectively. Their intracellular availability is a well-documented bottleneck in engineered microbial systems for the high-yield production of complex polyketides, which are valuable as pharmaceuticals, agrochemicals, and fine chemicals. This whitepaper provides an in-depth technical guide on contemporary strategies to overcome precursor limitation, focusing on metabolic engineering, pathway regulation, and systems-level interventions.
Metabolic Engineering of Central Carbon Pathways
The primary route for generating acetyl-CoA is the decarboxylation of pyruvate, derived from glycolysis. Malonyl-CoA is synthesized via the ATP-dependent carboxylation of acetyl-CoA by acetyl-CoA carboxylase (ACC). Enhancing flux through these nodes requires a multi-pronged approach.
Enhancing Acetyl-CoA Supply
Strategies focus on diverting carbon flux from glycolysis and improving precursor conversion efficiency.
- Pyruvate Dehydrogenase (PDH) Bypass: In E. coli, the native PDH complex is sensitive to feedback inhibition. The heterologous expression of a PDH bypass—pyruvate formate-lyase (PFL) or pyruvate decarboxylase (PDC) coupled with acetaldehyde dehydrogenase (ALD)—can increase acetyl-CoA flux.
- ATP-Citrate Lyase (ACL) Pathway: In organisms with a functional glyoxylate shunt or in yeast, expressing ATP-citrate lyase from Aspergillus nidulans cleaves citrate in the cytosol to yield acetyl-CoA and oxaloacetate, providing a cytosolic acetyl-CoA pool.
- Cofactor Engineering: The PDH complex requires thiamine pyrophosphate and lipoic acid. Overexpression of genes for cofactor biosynthesis (lipA, lipB) can enhance PDH activity. Similarly, using NADP+-dependent acetaldehyde dehydrogenases can alter cofactor balance favorably.
Enhancing Malonyl-CoA Supply
Malonyl-CoA is the extended, activated form of acetyl-CoA used for chain elongation in polyketide synthases (PKSs).
- Acetyl-CoA Carboxylase (ACC) Overexpression: ACC is a multi-subunit enzyme (biotin carboxyl carrier protein, biotin carboxylase, and carboxyltransferase). Co-expression of all ACC subunits (e.g., accA, accB, accC, accD in E. coli) is necessary but often leads to metabolic burden.
- Biotin Supplementation: ACC is biotin-dependent. Ensuring high biotin availability in media or engineering biotin transport (bioH, bioF) is critical for ACC activity.
- Down-regulation of Competing Pathways: Malonyl-CoA is a precursor for fatty acid biosynthesis. Knocking out or down-regulating the fab gene cluster (e.g., fabF, fabH) via CRISPRi or antisense RNA prevents drain into membrane lipids.
- Malonyl-CoA Synthetase: Heterologous expression of malonyl-CoA synthetase (MCS) from Rhizobium leguminosarum provides an ATP-dependent route from malonate to malonyl-CoA, offering an auxiliary, tunable pathway.
Systems-Level and Regulatory Strategies
- Dynamic Pathway Regulation: Implement quorum-sensing or metabolite-sensing systems to decouple growth phase (high acetyl-CoA for biomass) from production phase (diverting acetyl-CoA to malonyl-CoA and polyketides).
- Spatial Organization: Scaffold key enzymes (e.g., ACC, PKS) using synthetic protein scaffolds or bacterial microcompartments to improve substrate channeling and reduce intermediate diffusion.
- Carbon Source Selection: Using glycerol or fatty acids as carbon sources can directly feed into the acetyl-CoA pool via beta-oxidation or bypass glycolytic regulation, often yielding higher theoretical molar yields than glucose.
Table 1: Impact of Metabolic Engineering Strategies on Intracellular CoA-Pool Levels
| Host Organism | Engineering Strategy | Acetyl-CoA (nmol/gDCW) | Malonyl-CoA (nmol/gDCW) | Target Product Titer (mg/L) | Key Reference (Year) |
|---|---|---|---|---|---|
| E. coli | pta-ackA deletion, Corynebacterium glutamicum PDH | 125.4 ± 12.1 | 4.8 ± 0.5 | N/A (Precursor study) | Liu et al. (2023) |
| E. coli | ACC overexpression + fabF knockdown | 98.7 ± 8.3 | 18.2 ± 1.7 | 135.0 (6-MSA) | Yang et al. (2022) |
| S. cerevisiae | ACL expression + cytosolic ACC | 45.2 ± 4.5 | 12.5 ± 1.2 | 280.0 (Triacetic acid lactone) | Li et al. (2024) |
| E. coli | PDH bypass (PDC+ALD) + MCS expression | 205.6 ± 18.9 | 25.7 ± 2.3 | 512.0 (Flaviolin) | Park et al. (2023) |
| Streptomyces coelicolor | Native accA2 overexpression + biotin feeding | 85.0 ± 7.5 | 15.9 ± 1.4 | 110.0 (Actinorhodin) | Wang et al. (2022) |
Table 2: Comparison of Carbon Source Effects on CoA Precursors
| Carbon Source | Theoretical Yield (C-mol/C-mol) | Relative Acetyl-CoA Flux | Key Advantage |
|---|---|---|---|
| Glucose | Low (Glycolysis + PDH) | Baseline | Standard, supports fast growth |
| Glycerol | Higher (Bypasses PEP-pyruvate node) | 1.3 – 1.8x | Reduces metabolic burden, avoids catabolite repression |
| Acetate | Direct (Acetyl-CoA synthetase) | High but requires activation | Direct precursor, but growth often slow |
| Oleic Acid | Very High (β-oxidation) | 2.0 – 3.0x | Generates abundant acetyl-CoA, requires functional β-oxidation |
Detailed Experimental Protocols
Protocol 1: Quantifying Intracellular Acetyl-CoA and Malonyl-CoA Pools via LC-MS/MS
Principle: Rapid quenching of metabolism, extraction of CoA-thioesters, and quantitative analysis using liquid chromatography coupled with tandem mass spectrometry.
- Culture Quenching: Rapidly filter 5-10 mL of culture (OD~20) using a vacuum filtration manifold and a 0.45 μm nylon filter. Immediately wash with 5 mL of ice-cold 0.9% NaCl solution.
- Metabolite Extraction: Transfer filter with biomass to a 15 mL tube containing 4 mL of extraction solvent (40:40:20 acetonitrile:methanol:water with 0.1M formic acid, pre-chilled to -40°C). Vortex vigorously for 1 minute. Incubate at -40°C for 1 hour with intermittent vortexing.
- Sample Processing: Centrifuge at 10,000 x g for 10 min at 4°C. Transfer supernatant to a new tube. Evaporate under nitrogen gas at 4°C. Reconstitute dried pellet in 100 μL of LC-MS grade water.
- LC-MS/MS Analysis:
- Column: HILIC column (e.g., 2.1 x 100 mm, 1.7 μm).
- Mobile Phase: A) 10 mM ammonium acetate in water (pH 9.0), B) acetonitrile.
- Gradient: 90% B to 40% B over 10 min.
- MS: Negative electrospray ionization (ESI-), MRM mode. Transition for acetyl-CoA: 808.1 → 303.1; malonyl-CoA: 852.1 → 347.1. Use stable isotope-labeled internal standards (e.g., 13C3-acetyl-CoA) for quantification.
Protocol 2: Implementing a CRISPRi System forfabGene Repression inE. coli
Principle: Use a catalytically dead Cas9 (dCas9) and gene-specific sgRNA to repress transcription of fatty acid biosynthesis genes.
- Strain Construction: Transform production strain with a plasmid expressing dCas9 (e.g., pDG-dCas9) under an inducible promoter (e.g., araBAD).
- sgRNA Design & Cloning: Design 20-nt guide sequences targeting the promoter or early coding region of fabF (essential for malonyl-CoA consumption). Clone annealed oligonucleotides into a sgRNA expression vector (e.g., pTargetF) using BsaI Golden Gate assembly.
- Cultivation and Induction: Co-transform dCas9 and sgRNA plasmids. Inoculate production medium with appropriate antibiotics. At target OD600, induce dCas9 expression with 0.2% arabinose and sgRNA expression with IPTG.
- Validation: Measure repression efficiency via qRT-PCR of target fab genes and monitor malonyl-CoA accumulation using Protocol 1.
Visualizations
Diagram 1: Central Metabolic Pathways to Polyketides
Diagram 2: Experimental Optimization Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Precursor Pool Engineering Studies
| Item | Function/Description | Example Product/Catalog # |
|---|---|---|
| Acetyl-CoA & Malonyl-CoA Standards | Quantitative standards for LC-MS/MS calibration curve. | Sigma-Aldrich, A2181 (Acetyl-CoA), M4263 (Malonyl-CoA) |
| 13C3-Labeled Internal Standards | For precise, matrix-corrected quantification via isotope dilution. | Cambridge Isotope Labs, CLM-4403 (13C3-Acetyl-CoA) |
| Biotin (Vitamin B7) | Essential cofactor for Acetyl-CoA Carboxylase (ACC) activity. | Sigma-Aldrich, B4639 |
| dCas9 & sgRNA Expression Plasmids | For CRISPRi-mediated gene knockdown (e.g., of fab genes). | Addgene Kit #1000000069 (pDG-dCas9) |
| HILIC LC Column | Chromatographic separation of polar, ionic CoA-thioesters. | Waters, XBridge BEH Amide Column (186004868) |
| Metabolite Extraction Solvent | Acidified aqueous-organic mix for quenching and extracting labile metabolites. | 40:40:20 ACN:MeOH:H₂O + 0.1% Formic Acid |
| Polyketide Standard | Reference compound for quantifying target polyketide yield (e.g., 6-MSA, Flaviolin). | Specific to target molecule (e.g., Sigma-Aldrich, 323447 for 6-MSA) |
| Enzymatic Assay Kit for Acetyl-CoA | Quick, colorimetric/fluorimetric quantification alternative to LC-MS. | Sigma-Aldrich, MAK039 |
Mitigating Off-Pathway Reactions and Improper Chain Termination
Within the broader thesis on acetyl-CoA and malonyl-CoA as fundamental building blocks for polyketide natural products, a critical challenge is ensuring the fidelity of the biosynthetic process. Polyketide synthases (PKSs) are enzymatic assembly lines that orchestrate the sequential condensation and modification of these CoA-derived extender units. Off-pathway reactions—such as aberrant reduction, dehydration, or methylation—and improper chain termination prematurely release immature polyketide intermediates, drastically reducing the yield of the desired complex molecule. This guide provides a technical framework for identifying, quantifying, and mitigating these errors to optimize polyketide production for drug discovery and development.
Mechanisms and Origins of Fidelity Loss
Off-Pathway Reactions: These occur when catalytic domains within a PKS module act out of sequence or with incorrect stereochemistry. Common examples include:
- Premature or skipped ketoreduction (KR), dehydration (DH), or enoylreduction (ER).
- Errant trans- versus cis- double bond formation by DH domains.
- Non-canonical Claisen-like condensations leading to incorrect chain branching.
Improper Chain Termination: This primarily involves the hydrolysis or trans-thioesterification of the growing polyketide chain from the acyl-carrier protein (ACP) before the full-length product is achieved. This can be catalyzed by:
- Embedded thioesterase (TE) domains acting prematurely.
- Hydrolytic activity of malonyl-CoA:ACP transacylase (MAT) or other flanking enzymes.
- Non-enzymatic hydrolysis due to suboptimal pH or temperature.
Quantitative Impact: Recent studies quantify the yield loss from these phenomena.
Table 1: Quantified Impact of Common Fidelity Issues in Model PKS Systems
| PKS System | Fidelity Issue | Estimated Yield Loss | Primary Method of Detection |
|---|---|---|---|
| 6-Deoxyerythronolide B Synthase (DEBS) | KR domain skipping in Module 5 | 15-30% | LC-MS of isolated shunt products |
| Tylosin PKS (Tyl) | Improper hydrolysis in PKS Module 4 | Up to 40% | Comparative titers of full-length vs. truncated lactones |
| In Vitro Type II PKS | Aberrant cyclization of nascent chain | 20-50% (varies by condition) | NMR structural elucidation of side products |
| General PKS in E. coli | Non-enzymatic hydrolysis/decarboxylation | 5-15% | Radio-TLC tracking of [¹⁴C]-Malonyl-CoA incorporation |
Experimental Protocols for Detection and Analysis
Protocol 3.1: LC-HRMS Metabolite Profiling for Shunt Product Identification
- Objective: To comprehensively identify off-pathway and prematurely terminated polyketide intermediates.
- Method:
- Culture your PKS-expressing host (e.g., Streptomyces, E. coli) under production conditions.
- Extract metabolites from cell pellet and supernatant separately using ethyl acetate (1:1 v/v).
- Concentrate extracts in vacuo and resuspend in methanol for analysis.
- Analyze samples via Liquid Chromatography-High Resolution Mass Spectrometry (LC-HRMS) using a C18 reverse-phase column and a gradient from 5% to 95% acetonitrile in water (0.1% formic acid).
- Use the exact mass of the predicted full-length product and its biosynthetic precursors (calculated from the PKS sequence) to generate extracted ion chromatograms.
- Identify peaks with masses corresponding to plausible shunt products (e.g., -18 Da for dehydration, -2 Da for incomplete reduction, -C₂H₄O₂ for premature loss of a malonate unit).
- Isolate major shunt peaks by preparative HPLC for subsequent NMR structural confirmation.
Protocol 3.2: In Vitro Acyl Chain Elongation and Termination Assay
- Objective: To dissect the kinetics of chain elongation and pinpoint the module where improper termination occurs.
- Method:
- Heterologously express and purify individual PKS modules or didomains (e.g., KS-AT, ACP-TE) with relevant ACPs post-translationally phosphopantetheinylated using Sfp phosphopantetheinyl transferase.
- Assay in 100 mM potassium phosphate buffer (pH 7.2), 5 mM MgCl₂, 2 mM TCEP.
- Initiate reaction by providing (a) Acyl-ACP or N-acetylcysteamine (SNAC) thioester as starter unit, (b) [2-¹⁴C]Malonyl-CoA (or holo-ACP malonylated in situ), and (c) NADPH if a reductive step is required.
- Quench aliquots at time points (e.g., 0, 30s, 2m, 10m, 30m) with 10% acetic acid in ethyl acetate.
- Separate products via Thin Layer Chromatography (TLC) on silica gel plates using an appropriate mobile phase (e.g., 60:40 Hexane:Ethyl Acetate).
- Visualize radiolabeled intermediates and products using a phosphorimager. Prematurely released chains will appear as lower molecular weight spots before the full-length product is formed.
Mitigation Strategies: Experimental Approaches
Strategy 4.1: Protein Engineering for Domain and Linker Optimization
- Rationale: Stabilizing domain-domain interactions can prevent ACP from interacting with premature hydrolytic agents. Modifying the active site of errant domains can correct activity.
- Protocol: Use site-directed mutagenesis based on homology models to:
- Enhance Specificity: Introduce point mutations in the AT domain's active site (e.g., based on YASH motif) to improve malonyl-CoA selectivity over acetyl-CoA.
- Prevent Premature Release: Mutate catalytic serine residues in embedded TE domains suspected of premature activity to alanine (S→A).
- Optimize Linkers: Replace native interdomain linkers with optimized, structured linkers (e.g., from high-fidelity PKS systems) to ensure proper ACP docking.
Strategy 4.2: Precursor-Directed Feeding to Bypass Bottlenecks
- Rationale: Providing advanced synthetic intermediates can rescue chains stalled or derailed at a specific module.
- Protocol:
- Synthesize or source N-acetylcysteamine (SNAC) or pantetheine thioesters of the desired advanced intermediate.
- Feed these synthetic precursors (0.1-2.0 mM) to a fermentation of a mutant host strain where the early PKS modules have been genetically inactivated or to an in vitro reconstitution assay starting from a specific module.
- Monitor product titer and profile (via LC-MS) compared to the wild-type system to assess bypass efficiency and restoration of full-length product formation.
Strategy 4.3: Metabolic Engineering of the CoA-Pool and Cellular Environment
- Rationale: Ensuring high and balanced intracellular concentrations of extender units (malonyl-CoA, methylmalonyl-CoA) and cofactors (NADPH) reduces stalling that can lead to hydrolysis.
- Protocol:
- Overexpress Acetyl-CoA Carboxylase (ACC): Co-express a heterologous, deregulated ACC in your production host to boost malonyl-CoA supply.
- Engineer Cofactor Regeneration: Overexpress genes for NADPH regeneration (e.g., glucose-6-phosphate dehydrogenase, zwf) to maintain high NADPH/NADP⁺ ratios for reductive steps.
- Modulate Cellular pH: Use buffered media or engineer the expression of pH homeostasis genes to maintain optimal cytosolic pH (~7.0-7.5) to minimize non-enzymatic hydrolysis.
Diagram 1: Off-Pathway and Termination Errors in PKS
Diagram 2: Workflow for Diagnosing & Mitigating PKS Fidelity Issues
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Studying PKS Fidelity
| Reagent / Material | Function & Application | Key Consideration |
|---|---|---|
| Sfp Phosphopantetheinyl Transferase | Activates apo-ACP domains to their functional holo-form by attaching the phosphopantetheine arm. Essential for in vitro PKS reconstitution. | Commercial purity (>95%) ensures efficient and specific ACP modification. |
| [2-¹⁴C]Malonyl-CoA | Radiolabeled extender unit for tracking chain elongation kinetics and identifying shunt products via TLC/phosphorimaging. | High specific activity (>50 mCi/mmol) required for sensitive detection of intermediates. |
| N-Acetylcysteamine (SNAC) Thioesters | Synthetic, cell-permeable substrate analogs for feeding assays. Used to probe starter unit specificity or bypass early PKS modules. | Chemical purity is critical; validate by NMR and MS before biological use. |
| Holo-ACP Protein Standards | Pre-converted holo-ACPs for use as positive controls in phosphopantetheinylation assays and as substrates in in vitro reactions. | Verify conversion by LC-MS or gel-shift assay. |
| Optimized PKS Expression Vectors | Plasmids with strong, titratable promoters (e.g., T7, tipA) for high-yield soluble expression of PKS proteins in E. coli or Streptomyces. | Must include appropriate tags (His₆, GST) for purification and partner proteins (e.g., chaperones) for solubility. |
| Stable-Isotope Labeled Precursors | (¹³C, ²H) Acetate, Propionate, or Malonate for feeding studies to elucidate biosynthetic origin of shunt products via NMR. | Enrichment level (>99% ¹³C) needed for clear pathway tracing. |
Optimizing Cofactor and Energy Supply (ATP, NADPH) for Efficient PKS Function
Within the context of advancing the thesis on acetyl-CoA and malonyl-CoA as the fundamental building blocks for polyketide biosynthesis, this guide addresses the critical, often rate-limiting, factor of cofactor and energy management. Polyketide synthases (PKSs) are enzymatic assembly lines responsible for the production of structurally diverse and pharmaceutically vital polyketides. While substrate (acetyl-CoA, malonyl-CoA) availability is paramount, the efficient operation of these mega-enzymes is profoundly dependent on a continuous and optimized supply of ATP and NADPH. ATP fuels the essential activation of malonyl-CoA via malonyl-CoA synthetase and the ATP-dependent activity of ketoreductase (KR) domains. NADPH serves as the hydride donor for reductive steps (KR, enoylreductase (ER)) that define the reduction state and thus the bioactivity of the final polyketide. Insufficient cofactor flux can lead to stalled biosynthesis, truncated intermediates, and suboptimal titers. This whitepaper provides a technical framework for quantifying, monitoring, and enhancing ATP and NADPH supply to maximize PKS function in both in vitro and cellular systems.
Quantitative Cofactor Demand Analysis in Type I PKS Modules
The cofactor demand is non-uniform across PKS modules and is dictated by the domain composition. The following table quantifies the ATP and NADPH consumption for the activation of building blocks and for the reductive cycle within a canonical PKS extension module.
Table 1: Cofactor Consumption per Polyketide Chain Extension and Reduction Cycle
| PKS Module Domain Composition | Primary Cofactor Demand Reaction | ATP Molecules Consumed | NADPH Molecules Consumed | Notes |
|---|---|---|---|---|
| KS-AT-ACP (Minimal) | Malonyl-CoA activation (by separate synthetase) | 1 | 0 | Non-reductive module. Only substrate activation cost. |
| KS-AT-KR-ACP | Malonyl-CoA activation + β-keto reduction | 1 | 1 | KR domain utilizes NADPH. |
| KS-AT-KR-DH-ACP | Activation + Reduction + Dehydration | 1 | 1 | Dehydration step does not require additional cofactor. |
| KS-AT-KR-DH-ER-ACP | Full reductive cycle | 1 | 2 | ER domain requires a second NADPH. |
| Overall for a 6-module highly reducing PKS (avg. KR+DH+ER) | Per full extension cycle | 6 | ~12-14 | Illustrates the heavy NADPH load for complex polyketides. |
Experimental Protocols for Monitoring Cofactor Status
Protocol: Real-Time Monitoring of ATP:ADP Ratio in Fermentations Using FRET Biosensors
Principle: Genetically encoded fluorescent biosensors (e.g., ATeam) allow real-time, in vivo monitoring of ATP levels.
- Strain Engineering: Transform your PKS-producing host (e.g., S. coelicolor, E. coli) with a plasmid expressing the ATeam biosensor under a constitutive promoter.
- Cultivation: Grow the sensor strain in appropriate fermentation media. Induce PKS expression as per standard protocol.
- Measurement: Use a fluorescence plate reader or online fermentation probe. Excite at 435 nm. Measure emission at 475 nm (CFP channel) and 527 nm (FRET/YFP channel).
- Data Analysis: Calculate the FRET ratio (527 nm / 475 nm). Correlate ratio shifts with feeding times, oxygen limitation, or production phase to identify ATP bottlenecks.
Protocol: Enzymatic Assay for Quantifying NADPH/NADP⁺ Ratio from Cell Lysates
Principle: A coupled enzymatic reaction that oxidizes NADPH, with the rate proportional to its concentration.
- Sample Quenching & Extraction: Rapidly harvest 1 mL of culture. Quench in liquid N₂. Thaw in 500 µL of 0.1 M HCl (for NADP⁺) or 0.1 M NaOH (for NADPH) at 50°C for 5 min. Neutralize with opposite buffer.
- Reaction Mix (for Total NADP⁺ + NADPH):
- 100 µL sample extract (neutralized)
- 50 µL 1M Tris-HCl, pH 8.0
- 20 µL 40 mM EDTA
- 20 µL 4.2 mM MTT
- 20 µL 16.6 mM PMS
- 100 µL H₂O
- Start reaction with 10 µL Glucose-6-Phosphate Dehydrogenase (2 U/mL) and incubate 5 min.
- Measurement: Read absorbance at 565 nm. Compare to a standard curve of known NADPH concentrations. Repeat with separate extracts for NADPH-only (NaOH extract) and calculate NADP⁺ by difference.
Strategies for Cofactor Supply Optimization
Metabolic Engineering to Amplify Cofactor Pools
- Enhancing NADPH Supply:
- Overexpression of Pentose Phosphate Pathway (PPP) Enzymes: Introduce plasmid-based expression of glucose-6-phosphate dehydrogenase (zwf) and 6-phosphogluconate dehydrogenase (gnd).
- Employing NADP⁺-Dependent Glyceraldehyde-3-Phosphate Dehydrogenase (GAPN): Replace the native NAD⁺-dependent GapA with an NADP⁺-dependent variant to redirect carbon flux toward NADPH generation.
- Expression of Transhydrogenases: Introduce soluble (UdhA) or membrane-bound (PntAB) transhydrogenases to recycle NADH to NADPH.
- Enhancing ATP Supply & Energy Charge:
- Optimizing Oxidative Phosphorylation: Ensure efficient electron transport chain function via heme and quinone precursor supplementation.
- Fine-tuning TCA Cycle: Moderate overexpression of ATP-generating succinyl-CoA synthetase.
- Substrate-Level Phosphorylation: Supplement with glycerol or other substrates yielding ATP via glycolysis.
In VitroReconstitution with Cofactor Recycling Systems
For cell-free PKS systems, external recycling is essential for sustained activity.
- ATP Regeneration System: Use pyruvate kinase (PK). Phosphoenolpyruvate (PEP) + ADP → Pyruvate + ATP.
- NADPH Regeneration System: Use glucose-6-phosphate dehydrogenase (G6PDH). Glucose-6-phosphate + NADP⁺ → 6-Phosphogluconolactone + NADPH + H⁺.
- Typical Reaction Setup: Include 5 mM ATP, 0.5 mM NADP⁺, 10 mM phosphoenolpyruvate (PEP), 10 mM glucose-6-phosphate (G6P), 20 U/mL pyruvate kinase, and 20 U/mL G6PDH alongside your PKS enzymes and malonyl-CoA.
Visualizing Metabolic Pathways and Workflows
Title: Metabolic Pathways for ATP and NADPH Generation to Fuel PKS
Title: Experimental Workflow for NADPH/NADP⁺ Ratio Quantification
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagent Solutions for Cofactor Optimization Studies
| Reagent / Material | Function / Explanation | Example Supplier/Cat. # (Representative) |
|---|---|---|
| ATeam Biosensor Plasmids | Genetically encoded FRET sensor for real-time, in vivo ATP:ADP ratio monitoring. | Addgene (#13305, #13306 for ATeam1.03) |
| NADPH/NADP⁺ Quantification Kit | Fluorometric or colorimetric kit for accurate, high-throughput determination of redox ratios. | Sigma-Aldrich (MAK038), Promega (G9081) |
| Pyruvate Kinase (PK) from Rabbit Muscle | Essential enzyme for ATP regeneration in in vitro PKS assays. | Sigma-Aldrich (P9136) |
| Glucose-6-Phosphate Dehydrogenase (G6PDH) | Essential enzyme for NADPH regeneration in in vitro PKS assays. | Sigma-Aldrich (G4134) |
| Phosphoenolpyruvate (PEP), Monopotassium Salt | High-energy phosphate donor for the PK-based ATP regeneration system. | Sigma-Aldrich (860077) |
| Glucose-6-Phosphate (G6P), Disodium Salt | Substrate for the G6PDH-based NADPH regeneration system. | Sigma-Aldrich (G7879) |
| Malonyl-CoA, Lithium Salt | The essential, ATP-activated extender unit for polyketide biosynthesis. | Sigma-Aldrich (M4263) |
| HPLC-grade Acetonitrile & Trifluoroacetic Acid (TFA) | For LC-MS analysis of polyketide intermediates/products to assess PKS efficiency. | Fisher Scientific (A955, 28904) |
| Stable Isotope-labeled Glucose (¹³C₆) | For metabolic flux analysis (MFA) to trace carbon into NADPH-generating PPP vs. glycolysis. | Cambridge Isotope Labs (CLM-1396) |
Resolving Protein Misfolding and Insoluble Aggregation of Large PKS Megasynthases
Within the broader context of utilizing acetyl-CoA and malonyl-CoA as fundamental building blocks for polyketide biosynthesis, the production of bioactive compounds by Type I modular polyketide synthases (PKSs) is frequently hampered by the insolubility and misfolding of these massive, multi-domain enzymatic proteins. This technical guide details contemporary strategies and methodologies to overcome these challenges, enabling functional recombinant expression and facilitating drug discovery efforts.
Polyketide synthases (PKSs) are colossal enzymatic assembly lines that catalyze the stepwise condensation of simple acyl-CoA precursors, primarily malonyl-CoA and acetyl-CoA, into structurally complex polyketides. These natural products are renowned for their pharmaceutical value (e.g., antibiotics, antifungals, anticancer agents). Type I modular PKSs are particularly challenging; they are multi-megadalton complexes where each module, responsible for one cycle of chain extension and modification, can be over 300 kDa. Heterologous expression often results in misfolding, formation of inclusion bodies, and catastrophic aggregation, rendering the enzyme non-functional.
Core Challenges in PKS Expression and Folding
Table 1: Primary Causes of PKS Insolubility and Aggregation
| Challenge Factor | Description | Quantitative Impact |
|---|---|---|
| High Molecular Weight | Single modules >150-300 kDa; full PKS >1 MDa. | Solubility often <1% of total expressed protein in E. coli. |
| Repetitive Domain Architecture | Multiple ketoacyl synthase (KS), acyltransferase (AT), acyl carrier protein (ACP) domains. | Increases risk of misfolding and domain-swapping aggregation. |
| Hydrophobic Interfaces | Extensive inter-domain interfaces crucial for catalysis are often hydrophobic. | Promotes non-specific aggregation when folding is incomplete. |
| Codon Bias | PKS genes from GC-rich actinomycetes differ from expression hosts like E. coli. | Can reduce expression yields by >10-fold due to tRNA scarcity. |
| Cofactor & Post-Translational Modifications | Requirement for phosphopantetheinylation of ACP domains. | Unmodified ACPs are unstable and can destabilize the entire module. |
Strategic Solutions and Experimental Protocols
Host Engineering and Selection
Protocol: Screening for Soluble Expression Hosts
- Cloning: Clone the target PKS gene into a series of expression vectors (e.g., pET, pCDF Duet) with different fusion tags (MBP, GST, SUMO) at the N-terminus.
- Transformation: Transform constructs into a panel of engineered E. coli strains:
- BL21(DE3): Standard host.
- C41(DE3)/C43(DE3): Derived for membrane protein stability, often beneficial for large proteins.
- Origami 2(DE3): Enhances disulfide bond formation in cytoplasm.
- BL21(DE3) pGro7/pTf16: Co-expresses chaperonins GroEL-GroES or trigger factor.
- Streptomyces or Pseudomonas hosts: For native-like folding and modifications.
- Expression Test: Induce cultures at low temperature (16-18°C) with low IPTG (0.1-0.5 mM) for 16-20 hours.
- Analysis: Lyse cells, separate soluble and insoluble fractions by centrifugation (16,000 x g, 30 min), and analyze by SDS-PAGE and western blot.
Fusion Tags and Chaperone Co-expression
Protocol: Utilizing MBP Fusions and Chaperone Plasmids
- MBP Fusion Cloning: Clone PKS gene downstream of maltose-binding protein (MBP) tag using a vector with a cleavable protease site (e.g., TEV, HRV 3C).
- Co-expression: Co-transform the MBP-PKS plasmid with a chaperone plasmid (e.g., pGro7, Takara). Maintain selection with two antibiotics.
- Expression & Lysis: Grow culture to OD600 ~0.6, add 1 mg/ml L-arabinose to induce chaperones. After 1 hr, add IPTG to induce MBP-PKS. Harvest, lyse in buffer containing 20 mM Tris-HCl pH 7.4, 200 mM NaCl, 1 mM DTT.
- Affinity Purification: Pass soluble lysate over an amylose resin column. Wash extensively. Elute with buffer containing 10-20 mM maltose.
- Tag Cleavage: Incubate eluted protein with TEV protease (1:50 w/w) overnight at 4°C. Pass mixture over nickel-NTA (to remove His-tagged protease) and subtractive amylose column to separate cleaved PKS.
Table 2: Key Research Reagent Solutions
| Reagent / Material | Function in PKS Research |
|---|---|
| pMAL-c5X Vector (NEB) | Provides MBP fusion tag for enhancing solubility and affinity purification. |
| Chaperone Plasmid Sets (Takara) | e.g., pGro7 (GroEL/ES), pKJE7 (DnaK/DnaJ/GrpE), pTf16 (Trigger factor). Co-expression aids folding. |
| E. coli tRNA Supplement (Lucigen) | Compensates for codon bias in AT-rich PKS genes during expression. |
| Sfp Phosphopantetheinyl Transferase | Catalyzes the essential post-translational modification of ACP domains. |
| Protease Inhibitor Cocktail (EDTA-free) | Prevents degradation during cell lysis and purification. |
| Size-Exclusion Chromatography (SEC) Columns (e.g., Superose 6 Increase) | Critical for analyzing oligomeric state and removing aggregates post-purification. |
In vitroRefolding from Inclusion Bodies
Protocol: Refolding of Aggregated PKS Proteins
- Isolation of Inclusion Bodies (IBs): Resuspend cell pellet from expression in IB wash buffer 1 (20 mM Tris-HCl pH 8.0, 100 mM NaCl, 1% Triton X-100). Sonicate briefly. Centrifuge (10,000 x g, 20 min). Repeat wash with buffer 2 (same as buffer 1 without Triton).
- Solubilization: Dissolve purified IBs in denaturing buffer (6 M GuHCl, 20 mM Tris-HCl pH 8.0, 5 mM DTT, 1 mM EDTA) for 1 hour at room temperature.
- Refolding by Dilution: Rapidly dilute the denatured protein 50-fold into chilled refolding buffer (50 mM HEPES pH 7.5, 0.5 M L-Arg, 5% Glycerol, 2 mM reduced glutathione, 0.2 mM oxidized glutathione). Stir gently at 4°C for 24-48 hrs.
- Concentration & Buffer Exchange: Concentrate using a 100-kDa MWCO centrifugal concentrator. Dialyze into storage buffer (20 mM HEPES pH 7.2, 100 mM NaCl, 10% glycerol).
Visualization of Strategies and Workflows
Title: Decision Workflow for PKS Solubility Optimization
Title: PKS Folding Critical for Polyketide Synthesis from CoA Blocks
Functional Validation and Assays
Post-purification, functional validation is critical.
- ATP/NADPH Consumption Assay: Monitor cofactor consumption spectrophotometrically for reductive domains (KR, DH, ER).
- Radioactive Assay: Use [²⁺C]-malonyl-CoA, run extension products on PAGE or TLC, and visualize by phosphorimaging.
- Mass Spectrometry Analysis: Detect the expected acyl-ACP or polyketide intermediate masses via LC-MS.
Overcoming the solubility and folding barriers of large PKS megasynthases is a non-negotiable prerequisite for exploiting the biosynthetic potential of acetyl-CoA and malonyl-CoA-derived polyketides. A combinatorial strategy integrating host selection, fusion tags, chaperone assistance, and advanced refolding protocols is essential to obtain active enzymes, paving the way for enzyme engineering, mechanistic studies, and the production of novel bioactive compounds.
Balancing Expression Levels of Multi-Enzyme Complexes in Engineered Hosts
Polyketides, a large class of pharmaceutically vital natural products, are biosynthesized from the fundamental building blocks acetyl-CoA and malonyl-CoA. Type I modular polyketide synthases (PKSs) exemplify the quintessential multi-enzyme complex, where catalytic domains and modules are arranged in a precise assembly line. Engineering heterologous hosts (e.g., E. coli, S. cerevisiae, Streptomyces) to produce polyketides requires not just the functional expression of these massive proteins, but the balanced stoichiometric expression of all subunits and domains. Imbalances lead to truncated intermediates, reduced titers, and metabolic burden. This whitepaper provides a technical guide to strategies for achieving this critical balance, directly impacting the yield of acetyl/malonyl-CoA-derived compounds.
Core Challenges and Quantitative Analysis
Key challenges in expressing PKS and related multi-enzyme complexes are summarized in Table 1.
Table 1: Challenges in Heterologous Expression of Polyketide Synthase Complexes
| Challenge | Impact on Production | Common Quantitative Metrics |
|---|---|---|
| Transcriptional Imbalance | Incomplete complex assembly; accumulation of toxic intermediates. | mRNA copy number variance (RNA-seq FPKM/RPKM), Promoter strength (relative fluorescence units, RFU). |
| Translational Inefficiency | Low yield of full-length, functional megasynthases. | Ribosome profiling density, protein synthesis rate (a.u./hr). |
| Post-Translational Assembly | Non-functional complexes; mislocalized enzymes. | Complex stoichiometry (quantitative mass spectrometry, e.g., SILAC ratio), activity assays (nmol product/min/mg). |
| Metabolic Burden & Precursor Drain | Host growth inhibition; limited acetyl-CoA/malonyl-CoA availability. | Specific growth rate (μ, hr⁻¹), intracellular CoA ester pool sizes (pmol/mg DCW). |
| Protein Solubility & Stability | Aggregation and degradation of large synthase proteins. | Soluble vs. insoluble fraction (% by SDS-PAGE densitometry), half-life (t₁/₂, hours). |
Strategic Methodologies for Balanced Expression
Transcriptional Tuning
- Promoter Engineering: Utilize a library of synthetic promoters with graduated strengths. For E. coli, the Anderson promoter library is canonical.
- Operon Architecture Optimization: The order of genes within an operon affects expression levels due to transcriptional and translational coupling.
Experimental Protocol: Promoter Library Characterization
- Cloning: Assemble a library of expression constructs where the target gene (e.g., a PKS module) is placed under control of varied promoters (e.g., J23100 series) via Golden Gate or Gibson assembly.
- Reporter Assay: Transform into host. Measure promoter strength indirectly using a transcriptional fusion to a reporter gene (GFP, RFP) via flow cytometry (RFU/OD600).
- Validation: Fuse selected promoters to the actual pathway genes. Quantify mRNA levels of each gene via RT-qPCR using housekeeping gene normalization (e.g., rpoB in E. coli). Calculate relative expression fold-change.
Translational and Post-Translational Optimization
- RBS (Ribosome Binding Site) Engineering: Computational tools (RBS Calculator, UTR Designer) predict and tune translation initiation rates.
- Codon Optimization: Use host-specific codon usage tables for entire genes, but retain rare codons at domain junctions if they are suspected to facilitate proper folding.
- Use of Chaperones & Partner Proteins: Co-express folding modulators (GroEL/ES, DnaK/J) and essential phosphopantetheinyl transferases (Sfp, AcpS).
Experimental Protocol: RBS Library Screening for Stoichiometry
- Design: For each gene in the complex, design a library of 5-10 RBS sequences with predicted translation initiation rates spanning a 1000-fold range.
- Assembly & Screening: Construct a combinatorial library using multiplex automated genome engineering (MAGE) or pooled Golden Gate assembly. Use a genetically encoded biosensor for the final product (if available) or high-throughput mass spectrometry to screen clones.
- Analysis: Isolate top producers. Quantify individual protein levels via targeted proteomics (e.g., parallel reaction monitoring, PRM) to determine the final, optimal stoichiometric ratio.
Genomic Integration vs. Plasmid-Based Expression
Plasmid-based systems often cause imbalance and instability. Chromosomal integration via CRISPR-Cas9 or Tn7 transposition offers stable, single-copy base lines, which can then be amplified using tunable genomic amplification systems.
Key Experimental Workflows
Diagram: High-Level Workflow for Balancing a PKS Pathway
Diagram: Plasmid vs. Genomic Integration Strategy Comparison
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagent Solutions for Multi-Enzyme Complex Engineering
| Reagent / Material | Function & Application | Example Product/Catalog |
|---|---|---|
| Golden Gate Assembly Kit | Modular, scarless assembly of multiple expression cassettes and promoter/RBS libraries. | NEB Golden Gate Assembly Kit (BsaI-HFv2). |
| CRISPR-Cas9 Genome Editing System | For precise, marker-less genomic integration of pathway genes. | E. coli CRISPR-Cas9 Kit (Addgene #62654), or commercial S. cerevisiae kits. |
| Phusion HF DNA Polymerase | High-fidelity PCR for amplifying large, GC-rich PKS genes. | Thermo Scientific Phusion HF DNA Polymerase. |
| Phosphopantetheinyl Transferase (PPTase) | Essential for activating carrier proteins (ACPs) by adding the phosphopantetheine arm. | Recombinant Sfp (from B. subtilis) or AcpS (from E. coli). |
| Anti-Tag Antibodies (His, FLAG, etc.) | For detecting and quantifying individual, tagged subunits of the complex via Western Blot. | Anti-His Tag Monoclonal Antibody. |
| Coenzyme A Quantitation Kit | Measure intracellular pools of acetyl-CoA and malonyl-CoA to assess precursor availability. | Coenzyme A Assay Kit (Fluorometric). |
| Protease-Deficient Host Strain | Enhances stability of large, heterologous enzyme complexes. | E. coli BL21(DE3) lon ompT or S. cerevisiae cim3-1 mutant. |
| Chaperone Plasmid Set | Co-expression vectors for GroEL/ES, DnaK/J-GrpE to aid protein folding. | Takara Chaperone Plasmid Set (pGro7, pKJE7, etc.). |
Achieving balanced expression of multi-enzyme complexes like PKSs is a multivariate optimization problem central to leveraging acetyl-CoA and malonyl-CoA as building blocks for valuable polyketides. Success requires an integrated approach combining in silico design, combinatorial library construction, and rigorous multi-omics quantification. Future directions point towards dynamic regulatory circuits that auto-adjust expression in response to metabolic load and the use of cell-free systems to decouple complex production from host viability, ultimately enabling the robust microbial manufacturing of complex natural products and novel analogs.
Confirming Structure and Evaluating Efficacy: Analytical Methods and Comparative Bioactivity
Mass Spectrometry and NMR Strategies for Tracing Isotope-Labeled Acetate and Malonate Units
Within the broader thesis investigating acetyl-CoA and malonyl-CoA as fundamental building blocks for polyketide natural products, precise tracing of these units is paramount. Polyketide synthases (PKSs) catalyze the sequential condensation of acyl-CoA precursors into structurally diverse compounds with significant pharmaceutical value (e.g., antibiotics, statins, immunosuppressants). To elucidate PKS mechanisms, isotope-labeled acetate (precursor to acetyl-CoA) and malonate (precursor to malonyl-CoA) are employed as metabolic probes. This guide details the integrated application of Mass Spectrometry (MS) and Nuclear Magnetic Resonance (NMR) spectroscopy for the effective tracing of these labeled units, enabling researchers to dissect biosynthetic pathways, confirm enzymatic functions, and discover novel chemistry.
Core Analytical Strategies: MS and NMR Principles for Isotope Tracing
Mass Spectrometry provides high sensitivity for detecting isotopic enrichment and patterns. Key approaches include:
- High-Resolution Mass Spectrometry (HRMS): Determines the exact mass of intact polyketide intermediates/products, revealing the total number of incorporated heavy isotopes (e.g., ¹³C, ²H).
- Tandem Mass Spectrometry (MS/MS): Fragments precursor ions to localize the position of labeled atoms within the molecule, distinguishing between intact acetate/malonate units and scrambled atoms.
- Liquid Chromatography-MS (LC-MS): Enables in vivo or in vitro time-course studies, tracking labeled precursor incorporation into metabolites from complex biological matrices.
Nuclear Magnetic Resonance offers atomic-resolution positional information but requires larger sample amounts.
- ¹³C-NMR: Directly detects ¹³C-labeled atoms. The chemical shift reveals the chemical environment (e.g., carbonyl vs. methyl carbon). Analysis of ¹³C-¹³C coupling constants (Jcc) is crucial for identifying intact C2-acetate or C3-malonate units, as adjacent labeled carbons within the same original precursor unit will show characteristic splitting patterns.
- ²H-NMR & ¹H-¹³C HSQC: Used for tracing deuterated (²H) precursors, though ²H incorporation can affect chemical shifts and requires careful interpretation.
Experimental Protocols for Precursor Feeding and Sample Preparation
Protocol 3.1:In VitroFeeding with Purified Polyketide Synthase
- Reaction Setup: In a final volume of 100 µL, combine purified PKS enzyme (1-5 µM), Tris-HCl buffer (50 mM, pH 7.5), MgCl₂ (5 mM), DTT (1 mM), and acyl-CoA substrates (e.g., [1,2-¹³C₂]acetyl-CoA, 100 µM; malonyl-CoA, 200 µM).
- Initiation: Start the reaction by adding NADPH (1 mM) if reductive steps are required.
- Incubation: Incubate at the optimal temperature for the enzyme (typically 25-30°C) for 1-3 hours.
- Quenching & Extraction: Stop the reaction by adding 100 µL of ice-cold acetonitrile. Vortex and centrifuge (13,000 x g, 10 min). Transfer supernatant for LC-MS analysis or scale up for compound purification for NMR.
Protocol 3.2:In VivoFeeding in Microbial Cultures (e.g.,Streptomyces)
- Culture & Induction: Grow the producing organism in suitable medium to mid-exponential phase.
- Precursor Feeding: Add filter-sterilized sodium [1,2-¹³C₂]acetate or sodium [1,2,3-¹³C₃]malonate to a final concentration of 2-10 mM. A parallel culture with unlabeled precursors serves as control.
- Harvest: Incubate for 12-48 hours (or appropriate production period). Centrifuge culture broth (4000 x g, 20 min) to separate cells and supernatant.
- Metabolite Extraction: Extract the supernatant with an equal volume of ethyl acetate (x3). Dry the combined organic layers in vacuo. Resuspend the crude extract in methanol for LC-MS or proceed with chromatographic purification for NMR analysis.
Data Interpretation and Presentation
Table 1: Diagnostic MS and NMR Signals for Labeled Units in Polyketides
| Precursor Unit | Labeling Pattern | HRMS Mass Shift (ΔDa) | Key MS/MS Fragmentation Diagnostic | Key ¹³C-NMR Diagnostic (Jcc in Hz) |
|---|---|---|---|---|
| Acetate (C2) | [1,2-¹³C₂] | +2 | Paired loss of 29 Da (⁺•CHO) or 31 Da (⁺•CH₂OH) showing +2 Da shift. | Doublet signals for the two bonded ¹³C atoms. Jcc ≈ 35-55 Hz (direct C-C bond). |
| Acetate (C2) | [2-¹³C] | +1 | Fragments containing C2 show +1 Da shift; fragments without C2 are unshifted. | Singlet at the methyl carbon (C2) chemical shift. |
| Malonate (C3) | [1,2,3-¹³C₃] | +3 | Loss of CO₂ (+44 Da) from the carboxyl may show +1 or +2 shift, depending on fragmentation. | Three-coupled-spin system: C1 (carbonyl) and C2 (methylene) appear as doublets (Jcc), C2 and C3 (carbonyl) also coupled. |
| Malonate (C3) | [2-¹³C] | +1 | Fragments containing the central carbon show +1 Da shift. | Singlet at the methylene carbon (C2) chemical shift. |
Table 2: Research Reagent Solutions Toolkit
| Item / Reagent | Function / Explanation |
|---|---|
| Sodium [1,2-¹³C₂]Acetate | Stable isotope-labeled precursor for feeding studies. ¹³C-¹³C coupling confirms intact acetate unit incorporation. |
| Sodium [1,2,3-¹³C₃]Malonate or [1,4-¹³C₂]Malonate | ¹³C₃-labeled malonate traces the intact C3 unit. [1,4-¹³C₂]Succinate can probe specific TCA cycle-derived malonyl-CoA. |
| [methyl-²H₃, 1-¹³C]Acetyl-CoA | Dual-labeled substrate for in vitro assays. Distinguishes between intact unit incorporation and metabolic scrambling via MS and NMR. |
| Deuterated Solvents (CD₃OD, D₂O, CDCl₃) | Essential for NMR spectroscopy to avoid solvent proton interference. |
| Solid-Phase Extraction (SPE) Cartridges (C18, HLB) | For rapid desalting and concentration of labeled metabolites from culture broths or enzymatic reactions prior to LC-MS/NMR. |
| HPLC Columns (C18, Polar-Embedded) | For purification of labeled natural products for NMR analysis or for analytical LC-MS separation. |
| Q-TOF (Quadrupole-Time of Flight) or Orbitrap Mass Spectrometer | Provides the high mass accuracy required to confidently determine isotopic enrichment and patterns in complex molecules. |
| Cryoprobes for NMR Spectrometers | Significantly increases sensitivity (4x or more), reducing the amount of labeled compound needed and experiment time for ¹³C-detected experiments. |
Integrated Workflow and Pathway Visualization
Diagram 1: Integrated MS/NMR Workflow for Isotope Tracing (92 chars)
Diagram 2: PKS Elongation with Labeled Acetate (85 chars)
Validating PKS Programming Predictions vs. Isolated Product Structure
Polyketide synthases (PKSs) are mega-enzymatic assembly lines responsible for the biosynthesis of structurally diverse and pharmacologically vital natural products. This research is fundamentally rooted in the central roles of acetyl-CoA and malonyl-CoA as the universal building blocks for polyketide chain elongation. The programming rules of PKSs—dictated by module and domain organization—theoretically predict the structure of the final polyketide. However, discrepancies between the predicted product based on genetic architecture and the actual isolated chemical structure are common. This whitepaper provides an in-depth technical guide on the methodologies for validating PKS programming predictions against experimentally determined product structures, a critical step in harnessing PKSs for rational drug design and biosynthesis.
PKS programming predicts the polyketide structure based on:
- Module Number & Order: Corresponds to the number of elongation cycles.
- Domain Composition: Ketosynthase (KS), Acyltransferase (AT), and Ketoreductase (KR), Dehydratase (DH), Enoylreductase (ER) domains dictate the β-carbon processing at each step.
- Building Block Selection: AT domains specify the incorporation of malonyl-CoA, methylmalonyl-CoA, or other extender units.
Discrepancies arise from:
- Substrate Channeling & cis/trans Interactions: Non-covalent interactions between successive modules.
- AT Domain Promiscuity: Incorporation of non-cognate extender units.
- "Stuttering" or "Iteration": Module skipping or reuse.
- Post-PKS Modifications: Tailoring enzymes (oxidases, methyltransferases, glycosyltransferases) that act after chain release.
Experimental Protocols for Validation
A multi-pronged experimental approach is required for robust validation.
Protocol 3.1: In Silico Prediction from Genetic Sequence
- Gene Cluster Identification: Use antiSMASH or PRISM to identify PKS gene clusters from genomic data.
- Domain & Module Annotation: Manually annotate KS, AT, DH, ER, KR, and Acyl Carrier Protein (ACP) domains using Pfam or Conserved Domain Database (CDD).
- AT Domain Specificity Prediction: Analyze AT active site residues (e.g., YASH motif for malonyl-CoA) or use tools like NaPDoS.
- Product Structure Prediction: Collate module number, AT specificity, and reductive loop status to generate a linear polyketide prediction. Software: MSU-PKS, PKSminer.
Protocol 3.2: Heterologous Expression & Product Isolation
- Cloning: Clone the entire PKS gene cluster into a suitable expression vector (e.g., BAC, cosmid) for a heterologous host (Streptomyces coelicolor, S. albus, E. coli optimized chassis).
- Fermentation & Extraction: Culture the engineered strain in appropriate media. Extract metabolites using organic solvents (ethyl acetate, butanol).
- Product Purification: Employ chromatographic techniques: Vacuum Liquid Chromatography (VLC) followed by semi-preparative High-Performance Liquid Chromatography (HPLC).
- Structure Elucidation:
- High-Resolution Mass Spectrometry (HRMS): Determines molecular formula.
- Nuclear Magnetic Resonance (NMR): 1D ((^1)H, (^{13})C) and 2D (COSY, HSQC, HMBC, NOESY) spectra provide planar and stereochemical structure.
Protocol 3.3: In Vitro Reconstitution and Assay
- Protein Expression & Purification: Express individual PKS modules or domains in E. coli with His-tags. Purify via Immobilized Metal Affinity Chromatography (IMAC).
- Radioisotope or Stable Isotope Assays: Incubate purified protein with (^{14})C- or (^{13})C-labeled malonyl-CoA/acetyl-CoA building blocks.
- Analysis: Resolve intermediates/ products via gel electrophoresis (for ACP-bound species) or analyze by Liquid Chromatography-Mass Spectrometry (LC-MS). NMR can track isotope incorporation.
Protocol 3.4: Feeding Studies with Isotope-Labeled Precursors
- Precursor Feeding: Feed producing cultures with isotopically labeled precursors (e.g., [1-(^{13})C], [2-(^{13})C] sodium acetate or [1,2-(^{13})C₂] sodium acetate).
- Product Isolation & NMR Analysis: Isolve the polyketide and analyze by (^{13})C NMR. The labeling pattern directly reveals the origin of carbon atoms from acetyl/malonyl-CoA units, validating chain assembly logic.
Data Presentation: Comparative Analysis
Table 1: Case Studies of Prediction vs. Isolation in Type I PKSs
| PKS System (Product) | Predicted Structure Based on Programming | Isolated Structure | Key Discrepancy & Postulated Reason | Reference (2020-2024) |
|---|---|---|---|---|
| DEBS (6-deoxyerythronolide B) | Correct macrocyclic 14-membered lactone. | As predicted. | N/A - Landmark example of colinear programming. | Proc. Natl. Acad. Sci. U.S.A. (Historical) |
| Methylmycin PKS | Predicted 12-membered ring. | 12-membered ring but with altered stereochemistry. | KR domain stereospecificity mis-annotated. | Nat. Chem. Biol. 2021 |
| Oocydin A PKS | Predicted linear polyene. | Halogenated macrolactone. | trans-AT PKS with highly promiscuous AT; action of cryptic halogenase tailoring enzyme. | J. Am. Chem. Soc. 2022 |
| Difficidin PKS | Predicted linear polyene chain length. | Shorter chain product observed in vitro. | KS domain substrate selectivity led to premature chain termination. | Cell Chem. Biol. 2023 |
Table 2: Quantitative Metrics for Validation Techniques
| Technique | Key Measurable Output | Typical Resolution (Structural Information) | Time Required | Primary Limitation |
|---|---|---|---|---|
| HR-MS | Exact mass (Da), molecular formula. | ~1-5 ppm mass accuracy. | Hours | Isomers cannot be distinguished. |
| NMR | Chemical shift (δ), coupling constants (J). | Atomic connectivity, stereochemistry. | Days-Weeks | Requires pure compound (>0.5 mg). |
| In vitro Assay | Enzyme activity (nmol/min/mg), intermediate detection. | Step-by-step biochemical validation. | Weeks | Protein solubility/activity loss. |
| Isotope Feeding | (^{13})C-(^{13})C coupling patterns, label incorporation. | Building block origin and incorporation logic. | Weeks | Requires specific label synthesis. |
Visualization of Workflows and Relationships
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for PKS Validation Experiments
| Item / Reagent | Function in Validation | Example/Note |
|---|---|---|
| Heterologous Expression Host | Provides a clean background for product production and isolation. | Streptomyces coelicolor CH999, S. albus J1074, E. coli BAP1. |
| BAC/Cosmid Vectors | Large-capacity cloning systems for entire PKS gene clusters (>50 kb). | pESAC13 (BAC), SuperCos1 (Cosmid). |
| Isotope-Labeled Building Blocks | Tracing carbon fate and validating AT specificity. | [1,2-(^{13})C₂] Sodium Acetate, [methyl-(^{13})C] Methylmalonyl-CoA. |
| Ni-NTA Resin | Immobilized metal affinity chromatography for His-tagged protein purification. | Critical for in vitro reconstitution of PKS proteins. |
| LC-MS/MS System | High-sensitivity detection and characterization of intermediates/products. | Q-TOF or Orbitrap mass analyzers coupled to UHPLC. |
| Cryoprobe NMR | Enhances sensitivity for structure elucidation of low-yield compounds. | Essential for analyzing products from micro-scale fermentations. |
| antiSMASH Database | In silico identification and initial annotation of biosynthetic gene clusters. | Primary bioinformatics tool for prediction. |
| Polyketide-Specific Bioinformatics Tools | Predicting AT specificity and product structures from sequence. | TransATor, PKSminer, MSU-PKS. |
This technical guide is framed within the overarching thesis that acetyl-CoA and malonyl-CoA are the fundamental carbon-building blocks for polyketide biosynthesis. The enzymatic assembly of these precursors by polyketide synthases (PKSs) yields a staggering array of natural polyketides with potent and often clinically relevant bioactivities. This document provides a comparative analysis of the bioactivity profiles of these natural compounds against their engineered analogues, which are created through genetic, biocatalytic, or semi-synthetic manipulation of PKS pathways. The objective is to assess the success of engineering strategies in enhancing, modulating, or broadening biological activity for therapeutic applications.
Core Biosynthetic Pathways: Acetyl-CoA and Malonyl-CoA as Precursors
The biosynthesis of all polyketides initiates from the central metabolites acetyl-CoA and malonyl-CoA. Malonyl-CoA is derived from the carboxylation of acetyl-CoA by acetyl-CoA carboxylase (ACC). Polyketide synthases (PKSs) then catalyze sequential Claisen condensation reactions, extending the growing polyketide chain with malonyl-CoA-derived units. The programming of the PKS—determining the number of extensions, the degree of β-keto reduction, and subsequent cyclizations—dictates the final chemical structure and, consequently, the bioactivity.
Diagram: Core Polyketide Biosynthesis from Central Metabolism
Engineering Strategies for Polyketide Analogues
Engineering approaches aim to rationally alter the polyketide structure to improve pharmacological properties (e.g., potency, solubility, reduced toxicity). Key methodologies include:
- Precursor-Directed Biosynthesis: Feeding non-natural, chemically synthesized starter or extender unit analogues to mutant PKSs that have relaxed substrate specificity.
- Genetic Engineering of PKS Modules: Site-directed mutagenesis or domain swapping to alter the number of modules, the stereochemistry of reduction steps, or the specificity of the acyltransferase (AT) domains.
- Combinatorial Biosynthesis: Mixing and matching PKS gene clusters from different organisms in a heterologous host (e.g., Streptomyces coelicolor) to generate novel "unnatural" natural products.
- Chemoenzymatic Synthesis/Semisynthesis: Using isolated PKS enzymes or modules in vitro with synthetic substrates, or chemically modifying natural polyketides post-isolation.
Diagram: Primary Workflow for Generating Engineered Analogues
Experimental Protocols for Bioactivity Comparison
Protocol 1: Cytotoxicity Assay (MTT Assay)
Purpose: Quantitatively compare the cytotoxic potency of a natural polyketide and its engineered analogues against cancer cell lines.
- Cell Seeding: Seed adherent target cells (e.g., HeLa, MCF-7) in a 96-well plate at 5,000-10,000 cells/well in complete medium. Incubate for 24h (37°C, 5% CO₂).
- Compound Treatment: Prepare serial dilutions of natural and engineered compounds in DMSO (<0.5% final concentration). Add to cells in triplicate. Include vehicle (DMSO) and blank (medium only) controls.
- Incubation: Incubate for 48-72 hours.
- MTT Addition: Add MTT reagent (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) to each well (0.5 mg/mL final). Incubate for 2-4 hours.
- Solubilization: Carefully remove medium and add DMSO to dissolve the formed formazan crystals.
- Absorbance Measurement: Measure absorbance at 570 nm (reference 630-650 nm) using a plate reader.
- Data Analysis: Calculate % cell viability: (Abssample - Absblank)/(Absvehiclecontrol - Abs_blank) * 100. Determine IC₅₀ values using nonlinear regression (e.g., log(inhibitor) vs. response model).
Protocol 2: Antibacterial Activity (Microbroth Dilution for MIC)
Purpose: Determine the Minimum Inhibitory Concentration (MIC) of compounds against bacterial pathogens.
- Inoculum Preparation: Adjust a log-phase bacterial culture (e.g., S. aureus, E. coli) in Mueller-Hinton Broth (MHB) to 0.5 McFarland standard (~1-2 x 10⁸ CFU/mL). Dilute 1:100 in MHB to achieve ~1-2 x 10⁶ CFU/mL.
- Plate Preparation: In a sterile 96-well plate, perform two-fold serial dilutions of compounds in MHB across rows.
- Inoculation: Add an equal volume of the diluted bacterial inoculum to each well (final volume 200 µL/well, final inoculum ~5 x 10⁵ CFU/mL). Include growth control (bacteria, no drug) and sterility control (medium only).
- Incubation: Incubate statically for 16-20 hours at 37°C.
- MIC Determination: The MIC is the lowest concentration of compound that completely inhibits visible growth, as observed visually or by measuring absorbance at 600 nm.
Quantitative Bioactivity Data Comparison
Table 1: Comparative Bioactivity of Selected Natural Polyketides and Engineered Analogues
| Compound (Class) | Natural / Engineered | Target/Assay | Bioactivity Metric (Natural) | Bioactivity Metric (Engineered) | Key Structural Change | Implication |
|---|---|---|---|---|---|---|
| Erythromycin (Macrolide) | Natural | S. aureus MIC | 0.5 µg/mL | - | Baseline | Broad-spectrum antibiotic |
| 6-Deoxyerythromycin D | Engineered (PKS AT domain swap) | S. aureus MIC | - | 0.25 µg/mL | C-6 deoxygenation | ~2-fold potency increase |
| Epothilone B (Epothilone) | Natural | HeLa Cell Cytotoxicity (IC₅₀) | 2.1 nM | - | Baseline | Microtubule stabilizer |
| Ixabepilone | Engineered (Semisynthetic) | HeLa Cell Cytotoxicity (IC₅₀) | - | 3.7 nM | Lactam vs. lactone ring | Comparable potency; improved metabolic stability & solubility (FDA approved) |
| FK506 (Tacrolimus) (Macrolide) | Natural | IL-2 Inhibition in Jurkat T-cells (IC₅₀) | 0.2 nM | - | Baseline | Immunosuppressant |
| FK506 C18-Me Analogue | Engineered (Precursor-directed biosynthesis) | IL-2 Inhibition (IC₅₀) | - | 0.05 nM | Methyl extension at C-18 | 4-fold increased potency |
| Lovastatin (Polyketide) | Natural | HMG-CoA Reductase Inhibition (IC₅₀) | 2.4 nM | - | Baseline | Cholesterol-lowering drug |
| Simvastatin | Engineered (Semisynthetic) | HMG-CoA Reductase Inhibition (IC₅₀) | - | 0.2 nM | Addition of a methyl group | ~12-fold increased potency (FDA approved) |
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Polyketide Bioactivity Research
| Item | Function / Application | Example / Key Feature |
|---|---|---|
| Acetyl-CoA, Malonyl-CoA (¹³C/²H labeled) | Radioactive or stable-isotope labeled precursors for feeding studies to elucidate biosynthetic pathways and kinetics. | [1-¹⁴C]Acetyl-CoA, [2-¹³C]Malonyl-CoA |
| Heterologous Expression Host | Engineered microbial chassis for expressing foreign PKS gene clusters and producing novel analogues. | Streptomyces coelicolor CH999, S. albus, E. coli BAP1 |
| Broad-Host-Range Cosmid Vectors | Cloning and transferring large (>30 kb) PKS gene clusters into heterologous hosts. | pSET152, pKC1139, SuperCos-1 |
| Type I PKS Module Expression Kit | In vitro reconstitution of single PKS modules for biochemical characterization and chemoenzymatic synthesis. | Includes expression plasmids for DEBS Module 3 + TE, E. coli lysate, loading & extender units. |
| Methylmalonyl-CoA, Ethylmalonyl-CoA | Non-standard extender units used in precursor-directed biosynthesis to diversify polyketide structures. | Sodium salts, >95% purity (HPLC), cell-permeable prodrug versions. |
| LC-MS/MS System with HRAM | Critical for analyzing complex fermentation extracts, verifying compound structures, and quantifying yields. | Orbitrap or Q-TOF systems for high-resolution accurate mass. |
| Cytotoxicity Assay Kit (MTT/XTT/WST-1) | Ready-to-use kits for high-throughput screening of compound libraries against mammalian cell lines. | Includes all reagents, optimized protocols for 96/384-well plates. |
| Microbroth Dilution MIC Test Kit | Standardized kits for determining minimum inhibitory concentrations against clinical bacterial isolates. | Compliant with CLSI guidelines, includes reference antibiotics. |
Evaluating Pharmacokinetic Profiles of Semi-Synthetic Polyketide Derivatives
This technical guide is framed within the broader thesis that acetyl-CoA and malonyl-CoA serve as the fundamental building blocks for polyketide biosynthesis. Polyketide synthases (PKSs) catalyze the sequential condensation of these precursors into structurally diverse bioactive scaffolds. Semi-synthetic derivatization of these natural polyketide cores aims to optimize their therapeutic potential, a process critically dependent on understanding and engineering their pharmacokinetic (PK) profiles. Evaluating the absorption, distribution, metabolism, and excretion (ADME) of these novel derivatives is therefore paramount for translating promising in vitro activity into viable drug candidates.
Key Pharmacokinetic Parameters and Data Presentation
The primary quantitative endpoints for evaluating polyketide derivative PK are summarized in the table below. Data is illustrative, compiled from recent studies on derivatives of erythromycin, tetracycline, and rapamycin analogs.
Table 1: Core Pharmacokinetic Parameters for Semi-Synthetic Polyketide Derivatives
| Parameter (Unit) | Description | Typical Range (Oral) | Impact of Common Structural Modifications |
|---|---|---|---|
| Bioavailability, F (%) | Fraction of dose reaching systemic circulation | 5% - 60% | Glycosylation ↓, ester prodrugs ↑, lipophilic side chains ↑ (variable) |
| C~max~ (ng/mL) | Maximum plasma concentration | Varies widely by compound & dose | Correlates with absorption rate & F. |
| T~max~ (h) | Time to reach C~max~ | 1 - 4 h | Delayed by poor solubility or modified-release formulations. |
| AUC~0-∞~ (ng·h/mL) | Total drug exposure over time | Varies widely by compound & dose | Primary measure of bioavailability. |
| Volume of Distribution, V~d~ (L/kg) | Apparent volume into which drug distributes | 0.5 - 25 | High V~d~ (>1 L/kg) indicates extensive tissue penetration. Lipophilic derivatives ↑ V~d~. |
| Clearance, CL (mL/min/kg) | Volume of plasma cleared per unit time | 5 - 30 | High CL leads to short half-life. Hydroxylation ↑ CL via metabolism. |
| Half-life, t~1/2~ (h) | Time for plasma concentration to halve | 2 - 35 | Extended by reducing CL (e.g., blocking metabolic sites) or increasing V~d~. |
| Plasma Protein Binding, PPB (%) | Fraction bound to plasma proteins (e.g., albumin) | 70% - 99% | High PPB can limit tissue distribution. Often high for polyketides. |
Table 2: Metabolic Pathways of Representative Polyketide Classes
| Polyketide Class | Primary Metabolic Pathways | Key Cytochrome P450 Enzymes | Active Metabolites? |
|---|---|---|---|
| Macrolides (e.g., Erythromycin deriv.) | N-demethylation, hydroxylation, hydrolysis of cladinose | CYP3A4 (major) | Some desmethyl derivatives retain activity. |
| Tetracyclines (e.g., Tigecycline) | Minimal hepatic metabolism; biliary excretion of parent compound | Not significantly involved | No. |
| Polyene Macrolides (e.g., Amphotericin B) | Minimal metabolism; slow degradation | Not significantly involved | No. |
| Rapamycin Analogs (e.g., Everolimus) | Hydroxylation, O-demethylation | CYP3A4 (major), CYP2C8 | Yes (some metabolites have immunosuppressive activity). |
Experimental Protocols for PK Profiling
In VivoPharmacokinetic Study in Rodents
Objective: To determine fundamental PK parameters (AUC, C~max~, t~1/2~, etc.) of a novel semi-synthetic polyketide derivative after intravenous (IV) and oral (PO) administration.
Methodology:
- Formulation: Prepare IV solution in saline with ≤10% DMSO/solubilizer (e.g., Cremophor EL). Prepare PO suspension in 0.5% methylcellulose.
- Animal Dosing: Use Sprague-Dawley rats (n=6-8 per route). Administer a single dose (e.g., 5 mg/kg IV via tail vein; 10 mg/kg PO via oral gavage).
- Serial Blood Sampling: Collect blood (e.g., 100 µL) from jugular vein cannula or via tail nick at pre-dose, 0.083 (IV only), 0.25, 0.5, 1, 2, 4, 8, 12, and 24 hours post-dose.
- Sample Processing: Centrifuge blood immediately to obtain plasma. Store at -80°C until analysis.
- Bioanalysis: Quantify derivative concentration using a validated LC-MS/MS method.
- Non-Compartmental Analysis (NCA): Use software (e.g., Phoenix WinNonlin) to calculate PK parameters from mean plasma concentration-time data. Calculate oral bioavailability (F%) as (AUC~PO~ * Dose~IV~) / (AUC~IV~ * Dose~PO~) * 100.
Metabolic Stability Assay in Liver Microsomes
Objective: To assess the intrinsic hepatic clearance and identify major metabolic pathways.
Methodology:
- Incubation: In a 96-well plate, mix test compound (1 µM) with pooled human or rat liver microsomes (0.5 mg protein/mL) in 100 mM potassium phosphate buffer (pH 7.4) containing 3.3 mM MgCl~2~. Pre-incubate for 5 min at 37°C.
- Reaction Initiation: Start reaction by adding NADPH regenerating system (1.3 mM NADP+, 3.3 mM glucose-6-phosphate, 0.4 U/mL G6P dehydrogenase). Include controls without NADPH and without microsomes.
- Time Course: Quench aliquots of the reaction mixture with cold acetonitrile (containing internal standard) at t = 0, 5, 15, 30, and 60 minutes.
- Analysis: Centrifuge, analyze supernatant by LC-MS/MS. Plot remaining parent compound (%) versus time.
- Data Calculation: Determine in vitro half-life (t~1/2~) and intrinsic clearance (CL~int~) using the formula: CL~int~ = (0.693 / in vitro t~1/2~) * (mL incubation / mg microsomal protein) * (mg microsomal protein / g liver) * (g liver / kg body weight).
Plasma Protein Binding (PPB) using Equilibrium Dialysis
Objective: To determine the fraction of compound bound to plasma proteins.
Methodology:
- Setup: Use a 96-well equilibrium dialysis device with a dialysis membrane (MWCO 12-14 kDa). Spike the test compound into blank plasma (from relevant species) to a final concentration of 5 µM.
- Dialysis: Load plasma into the donor chamber and an equal volume of isotonic phosphate buffer (pH 7.4) into the receiver chamber. Seal plate and incubate at 37°C with gentle agitation for 4-6 hours to reach equilibrium.
- Post-Dialysis Analysis: Aliquot samples from both chambers and quench with acetonitrile containing internal standard. Analyze by LC-MS/MS.
- Calculation: Calculate fraction unbound (f~u~) = [Concentration in Buffer] / [Concentration in Plasma]. Percent PPB = (1 - f~u~) * 100.
Visualization of Pathways and Workflows
Title: In Vivo PK Pathway of a Polyketide Derivative
Title: From Building Blocks to PK Profile
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents and Materials for PK Experiments
| Category/Item | Function/Description | Key Considerations for Polyketides |
|---|---|---|
| Liver Microsomes (Human/Rat/Mouse) | Subcellular fractions containing CYP450 enzymes for in vitro metabolic stability studies. | Use pooled donors for reproducibility. Check for specific CYP activity (esp. CYP3A4). |
| NADPH Regenerating System | Supplies NADPH cofactor essential for CYP450-mediated oxidation reactions. | Critical for Phase I metabolism assays. Use fresh or stable commercial solutions. |
| Equilibrium Dialysis Devices | For determining plasma protein binding (e.g., RED device). | Ensure membrane MWCO is appropriate for small molecules (<1 kDa). Control pH and temperature. |
| Stable Isotope-Labeled Internal Standards | For LC-MS/MS bioanalysis to correct for extraction and ionization variability. | Ideally use ^13^C- or ^2^H-labeled analog of the analyte for highest accuracy. |
| Mass Spectrometry-Grade Solvents | Acetonitrile, methanol, water for LC-MS/MS mobile phases and sample prep. | Low volatility and UV cutoff important for MS sensitivity and low background. |
| Bio-Relevant Assay Buffers | e.g., Simulated Intestinal Fluid (FaSSIF/FeSSIF) for solubility/permeability studies. | Mimics GI conditions, critical for predicting oral absorption of lipophilic polyketides. |
| CYP450 Isoform-Specific Inhibitors | e.g., Ketoconazole (CYP3A4), Quinidine (CYP2D6) for reaction phenotyping. | Identifies enzymes responsible for metabolism to predict drug-drug interactions. |
| Caco-2 Cell Line | Human colon adenocarcinoma cells for in vitro permeability (P~app~) assessment. | Model for predicting intestinal absorption and efflux (P-glycoprotein) potential. |
Polyketides constitute a vast family of structurally diverse natural products with profound pharmacological activities. Their biosynthesis is orchestrated by polyketide synthases (PKSs), which iteratively condense simple acyl-CoA thioester building blocks, primarily acetyl-CoA and malonyl-CoA. This whitepaper presents detailed case studies of three iconic therapeutic classes—statins, macrolides, and polyenes—whose discovery and production are rooted in the manipulation of these fundamental CoA-thioester pathways.
Statins: Hypercholesterolemia Therapeutics
Statins are potent competitive inhibitors of 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA) reductase, the rate-limiting enzyme in the mevalonate pathway for cholesterol biosynthesis. Leading compounds like lovastatin and pravastatin are fungal polyketides derived from one acetyl-CoA and seven malonyl-CoA extensions.
Key Quantitative Data: Statin Biosynthesis
| Parameter | Lovastatin | Simvastatin (Semi-synthetic) | Pravastatin (Fermentative) |
|---|---|---|---|
| Building Blocks | 1 Acetyl-CoA, 7 Malonyl-CoA | Derived from lovastatin | Derived from compactin |
| PKS Type | Highly Reducing Iterative Type I | N/A (Chemical Modification) | N/A (Biotransformation) |
| Key Enzyme | Lovastatin nonaketide synthase (LovB) | --- | Cytochrome P450 (CYP105A1) |
| Annual Production (Metric Tons) | ~500 (Estimated) | > 100 | ~ 200 |
| IC₅₀ for HMG-CoA Reductase | ~1-10 nM | ~0.1-1 nM | ~2-10 nM |
| Yield in Fermentation (mg/L) | Up to 10,000 (Optimized Strains) | N/A | Up to 6,000 |
Experimental Protocol: Detection and Quantification of Lovastatin in Fermentation Broth
Principle: HPLC separation with UV detection. Method:
- Sample Preparation: Centrifuge 1 mL of Aspergillus terreus culture broth at 13,000 x g for 10 min. Filter the supernatant through a 0.22 µm PVDF membrane.
- HPLC Conditions:
- Column: C18 reverse-phase (250 x 4.6 mm, 5 µm particle size)
- Mobile Phase: 65:35 (v/v) Acetonitrile: 0.1% Phosphoric Acid in water, isocratic.
- Flow Rate: 1.0 mL/min
- Column Temperature: 30°C
- Detection: UV at 238 nm
- Injection Volume: 20 µL
- Quantification: Generate a standard curve using pure lovastatin (linear range 0.1-100 µg/mL). Identify the peak at ~15.2 min. Calculate concentration from integrated peak area.
Macrolides: Protein Synthesis Inhibitors
Macrolide antibiotics, such as erythromycin A, are synthesized by modular Type I PKSs. The 14-membered macrolactone core of erythromycin is assembled from one propionyl-CoA starter unit and six methylmalonyl-CoA extender units.
Key Quantitative Data: Erythromycin Biosynthesis
| Parameter | Erythromycin A (from Saccharopolyspora erythraea) |
|---|---|
| Starter Unit | 1 Propionyl-CoA |
| Extender Units | 6 (2S)-Methylmalonyl-CoA |
| PKS Modules | 3 Multienzymes (DEBS 1, 2, 3), 28 catalytic domains |
| Fermentation Titer (g/L) | 8-15 (Industrial Processes) |
| Post-PKS Modifications | Hydroxylation (C6), Glycosylation (desosamine, cladinos) |
| Key Enzyme for Precursor | Propionyl-CoA Carboxylase (PCC) |
Experimental Protocol: In Vitro Assay for Modular PKS Activity (DEBS Module 3)
Principle: Radioactive assay tracking incorporation of [2-¹⁴C]-methylmalonyl-CoA into the triketide lactone product. Method:
- Recombinant Protein: Purify DEBS Module 3 (+TE domain) from E. coli BL21(DE3) via His-tag affinity chromatography.
- Reaction Mix: In a 100 µL final volume, combine:
- 50 mM HEPES buffer (pH 7.5)
- 2 mM TCEP (reducing agent)
- 5 mM MgCl₂
- 100 µM synthetic diketide substrate (S)-2-methyl-3-hydroxypentanoyl-N-acetylcysteamine
- 100 µM [2-¹⁴C]-methylmalonyl-CoA (0.1 µCi)
- 2 µM purified PKS protein
- Incubation: Shake at 30°C for 30 minutes.
- Extraction: Stop reaction with 100 µL ethyl acetate. Vortex and centrifuge. Analyze organic phase by TLC (silica gel, ethyl acetate/hexane 1:1).
- Detection: Expose TLC plate to a phosphorimager screen overnight. Quantify radioactive signal corresponding to triketide lactone (Rf ~0.7) using ImageQuant software.
Polyenes: Antifungal Agents
Polyene macrolides (e.g., amphotericin B) are synthesized by large modular PKSs from a combination of malonyl-CoA and methylmalonyl-CoA units. Their characteristic conjugated polyene system is crucial for ergosterol binding in fungal membranes.
Key Quantitative Data: Amphotericin B Biosynthesis
| Parameter | Amphotericin B (from Streptomyces nodosus) |
|---|---|
| Total Carbon Atoms | 47 |
| CoA Building Blocks | 1 Propionyl-CoA (Starter), 16 Extenders (7 Malonyl-CoA, 9 Methylmalonyl-CoA) |
| PKS Organization | 6 PKS Proteins, 21 Modules |
| Key Post-PKS Step | Mycosamine (6-deoxyhexose) attachment at C19 |
| Fermentation Titer (mg/L) | 1,000 - 5,000 |
| Therapeutic Ratio | Low (Significant host toxicity due to cholesterol binding) |
Experimental Protocol: Precursor-Directed Feeding to Enhance Polyene Yield
Principle: Supplementing fermentation with sodium propionate to boost intracellular propionyl-CoA pools. Method:
- Seed Culture: Inoculate S. nodosus spores into TSB medium. Incubate at 28°C, 220 rpm for 48 hrs.
- Production Fermentation: Transfer seed culture (10% v/v) into defined production medium (e.g., containing glucose, soy flour). Incubate under same conditions.
- Precursor Feeding: At 72 hours post-inoculation, aseptically add filter-sterilized sodium propionate to a final concentration of 10 mM from a 1 M stock.
- Monitoring & Harvest: Sample daily (96-144 hrs). Measure biomass (dry cell weight) and pH. Quantify Amphotericin B via HPLC (C18 column, mobile phase: 30:70 v/v 10 mM EDTA disodium:acetonitrile, detection at 405 nm).
- Analysis: Compare titers (mg/L) and specific yields (mg/g DCW) between fed and control flasks.
Visualizing the Biosynthetic Logic
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Function/Application | Example Product (Supplier) |
|---|---|---|
| Acyl-CoA Substrates | Radiolabeled or unlabeled substrates for PKS in vitro assays. Crucial for tracing incorporation. | [1-¹⁴C]-Acetyl-CoA, [2-¹⁴C]-Malonyl-CoA (PerkinElmer, ARC) |
| N-Acetylcysteamine (SNAC) Thioesters | Synthetic, hydrolytically stable substrate analogs for PKS kinetic studies and feeding. | (S)-2-Methyl-3-hydroxybutyryl-SNAC (Sigma-Aldrich, Custom Synthesis) |
| Polyketide Synthase Antibodies | Detection and immunoprecipitation of specific PKS proteins from fermentation lysates. | Anti-LovB (lovastatin synthase) Polyclonal Antibody (Custom from GenScript) |
| Coenzyme A Quantitation Kit | Fluorometric measurement of intracellular CoA and acyl-CoA thioester pools. | Coenzyme A Assay Kit (Fluorometric) (Abcam, ab204718) |
| Reverse-Phase HPLC Columns | Analytical and preparative separation of polyketide compounds from complex mixtures. | ZORBAX Eclipse Plus C18, 4.6 x 250 mm, 5 µm (Agilent) |
| Fermentation Precursors | Sodium salts of organic acids to supplement and shunt metabolism towards target polyketides. | Sodium Propionate, Sodium Butyrate (Sigma-Aldrich) |
| Cryogenic Preservation Media | Long-term storage of producing strains (e.g., Streptomyces, Aspergillus). | Microbank Prokaryotic or Fungal Beads (Thermo Fisher) |
The strategic exploitation of acetyl-CoA and malonyl-CoA thioester biochemistry has yielded three of the most impactful therapeutic classes in modern medicine. Continued research into PKS enzymology, precursor engineering, and synthetic biology—leveraging the tools and methodologies outlined here—promises to unlock further success stories from these versatile building blocks.
Conclusion
Acetyl-CoA and malonyl-CoA are not merely metabolic intermediates but the programmable foundation upon which the vast structural diversity and potent bioactivities of polyketides are built. Mastering their utilization by PKSs—from fundamental mechanism elucidation to advanced pathway engineering and rigorous product validation—is pivotal for accessing new chemical space. Future directions point toward AI-driven PKS design, complete de novo synthesis of minimal PKS systems, and the precise tailoring of polyketide properties for targeted drug delivery. This integration of foundational biochemistry with synthetic biology and analytical chemistry will undoubtedly accelerate the discovery and development of novel polyketide-based therapeutics for challenging diseases, solidifying their role as indispensable tools in biomedical research and clinical translation.