Domain Swapping Strategies in NRPS Engineering: A Comprehensive Guide for Novel Antibiotic Discovery
This article provides a detailed exploration of Nonribosomal Peptide Synthetase (NRPS) domain swapping, a cornerstone strategy in synthetic biology for engineering novel bioactive compounds.
Domain Swapping Strategies in NRPS Engineering: A Comprehensive Guide for Novel Antibiotic Discovery
Abstract
This article provides a detailed exploration of Nonribosomal Peptide Synthetase (NRPS) domain swapping, a cornerstone strategy in synthetic biology for engineering novel bioactive compounds. We systematically cover the foundational architecture and logic of NRPS mega-enzymes, then delve into cutting-edge methodologies for precise domain and module exchange, including split-intein and advanced cloning techniques. The guide addresses critical troubleshooting and optimization challenges, such as maintaining protein solubility and linker/junction compatibility. Finally, we evaluate validation frameworks and compare domain swapping to alternative engineering approaches, offering researchers and drug development professionals a practical, state-of-the-art resource for reprogramming nature's assembly lines to combat antimicrobial resistance.
Decoding the Blueprint: Understanding NRPS Architecture for Effective Domain Swapping
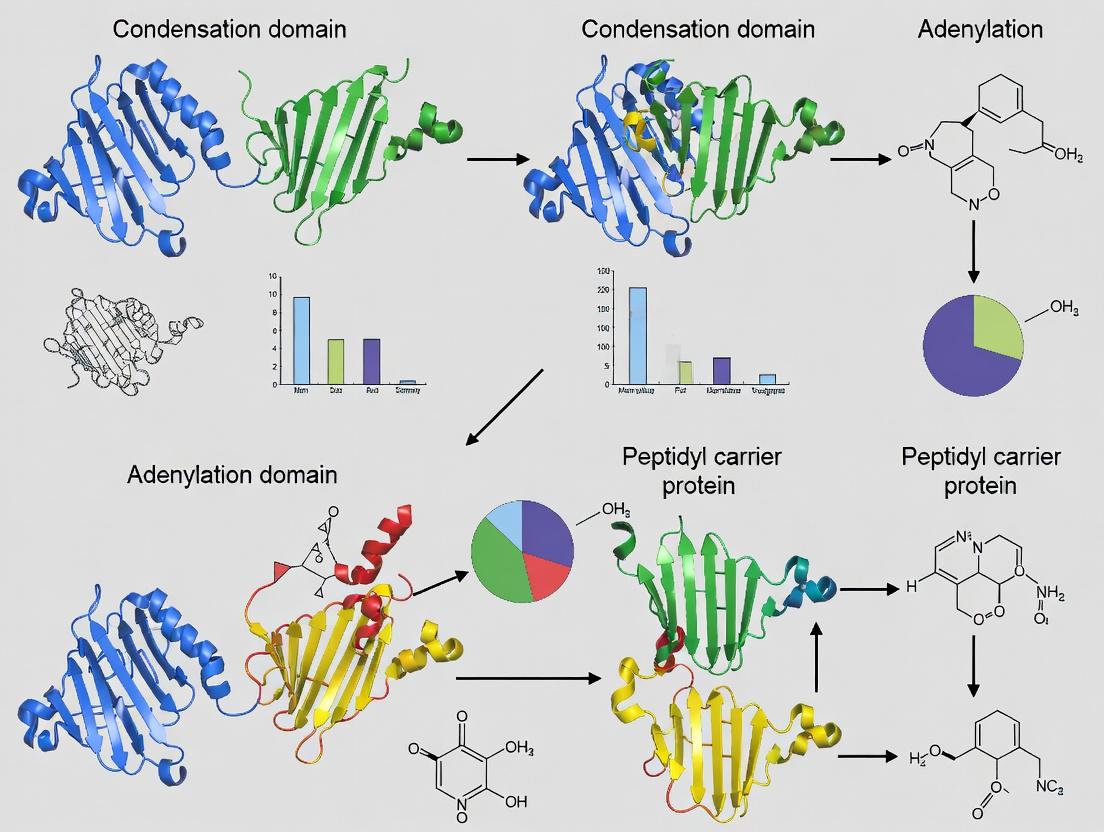
Nonribosomal peptide synthetases (NRPSs) are multi-modular enzymatic assembly lines responsible for the biosynthesis of numerous bioactive peptides with pharmaceutical importance, including antibiotics (penicillin, vancomycin), immunosuppressants (cyclosporine), and anticancer agents (bleomycin). The core architecture of a canonical NRPS elongation module consists of three essential domains: Adenylation (A), Thiolation (T), and Condensation (C). These domains function in a coordinated, assembly-line fashion to activate, thioesterify, and condense amino acid building blocks. Within the context of NRPS engineering for novel compound production, domain swapping—the exchange of these core domains between different NRPS systems—represents a primary rational strategy. This document provides application notes and detailed experimental protocols for studying and manipulating these fundamental building blocks, serving as a methodological resource for thesis research on domain swapping strategies.
Core Domains: Functions & Quantitative Parameters
The table below summarizes the key functions and biochemical parameters of the three core NRPS domains.
Table 1: Core NRPS Domain Functions and Characteristics
| Domain | Core Function | Key Biochemical Step | Essential Motifs/Co-factors | Typical Size (kDa) | Catalytic Rate (kcat, min⁻¹) Example |
|---|---|---|---|---|---|
| Adenylation (A) | Substrate recognition and activation | ATP-dependent aminoacyl-AMP formation; aminoacyl transfer to T domain. | A4, A7, A10 core motifs; Mg²⁺ | ~55-70 | 50-200 (e.g., PheA: ~120) |
| Thiolation (T) / Peptidyl Carrier Protein (PCP) | Activated intermediate carrier | Covalent binding of amino acid/peptide via thioester linkage to its prosthetic group. | Conserved serine for 4'-phosphopantetheine (PPant) arm attachment; holo-form required. | ~10-12 | N/A (Carrier) |
| Condensation (C) | Peptide bond formation | Catalyzes nucleophilic attack of the incoming aminoacyl-/peptidyl-T on the upstream peptidyl-T. | HHxxxDG motif (His-histidine brace), DXXXXD motif; critical for stereoselectivity. | ~50 | 5-30 (e.g., VibH: ~8) |
Experimental Protocols for Domain Analysis and Swapping
Protocol: In Vitro Adenylation (A) Domain Activity Assay (ATP-PPi Exchange)
Objective: To quantitatively measure the substrate specificity and activation kinetics of an A domain. Principle: The A domain catalyzes the reversible formation of aminoacyl-AMP and pyrophosphate (PPi). Radiolabeled [³²P]PPi is incorporated into ATP in the presence of the correct amino acid substrate, providing a measurable activity.
Materials:
- Purified A domain or NRPS module protein.
- Candidate amino acid substrates.
- [³²P]PPi (e.g., PerkinElmer NEG027).
- ATP, MgCl₂.
- Reaction Buffer: 50 mM Tris-HCl (pH 7.5), 10 mM MgCl₂, 5 mM DTT, 1 mM EDTA.
- Charcoal Suspension: 4% (w/v) activated charcoal in 0.1 M HCl, 1 mM Na₄P₂O₇.
- Scintillation counter.
Procedure:
- Reaction Setup: For each amino acid test, prepare a 100 µL reaction mix containing: 50 mM Tris-HCl (pH 7.5), 10 mM MgCl₂, 5 mM DTT, 1 mM EDTA, 5 mM ATP, 2 mM amino acid, 1 mM [³²P]PPi (~0.1 µCi), and 0.1-1 µM purified enzyme.
- Incubation: Incubate at 25-30°C for 10-30 minutes.
- Termination & Binding: Stop the reaction by adding 1 mL of ice-cold charcoal suspension. Vortex and incubate on ice for 10 min. The charcoal binds nucleotide phosphates (ATP).
- Washing: Centrifuge at 13,000 rpm for 5 min at 4°C. Carefully aspirate the supernatant. Wash the charcoal pellet twice with 1 mL of distilled water.
- Measurement: Resuspend the final pellet in 0.5 mL water and transfer to a scintillation vial with cocktail. Measure radioactivity (CPM) via scintillation counting.
- Analysis: Plot CPM vs. time or substrate concentration. Calculate kinetic parameters (Km, kcat) after correcting for background (no-enzyme control).
Protocol: Holo-Form Generation of T Domains using Phosphopantetheinyl Transferase (PPTase)
Objective: To convert the inactive apo-T domain (lacking the PPant arm) to the active holo-form. Principle: A broad-spectrum PPTase (e.g., Sfp from Bacillus subtilis) transfers the PPant moiety from coenzyme A (CoA) to the conserved serine of the T domain.
Materials:
- Purified apo-T domain or multi-domain protein containing T domains.
- Sfp PPTase (commercial, e.g., Sigma-Aldrich, or purified).
- Coenzyme A (CoA) or acyl-CoA derivatives.
- Reaction Buffer: 50 mM HEPES (pH 7.5), 10 mM MgCl₂.
- HPLC or mass spectrometry system for analysis.
Procedure:
- Reaction: In a total volume of 100 µL, combine: 50 mM HEPES (pH 7.5), 10 mM MgCl₂, 50 µM apo-protein, 100 µM CoA, and 0.5 µM Sfp PPTase.
- Incubation: Incubate at 30°C for 1-2 hours.
- Verification: Analyze the reaction product by LC-MS or a gel-shift assay (holo-proteins often migrate slightly slower in non-denaturing PAGE). A successful reaction is indicated by a mass increase of 340 Da (for CoA) or the corresponding acyl-group mass.
- Purification: If needed, remove excess CoA and PPTase by desalting or affinity chromatography.
Protocol: Two-ModuleIn VitroReconstitution Assay for C Domain Activity
Objective: To directly test the condensation activity and donor-acceptor specificity of a C domain. Principle: Two NRPS modules are loaded with their cognate amino acids (donor module as peptidyl-S-T, acceptor module as aminoacyl-S-T). The C domain catalyzes peptide bond formation only if it recognizes the correct upstream and downstream intermediates.
Materials:
- Purified donor and acceptor modules (minimally, C-A-T and A-T or T didomain constructs).
- Required A domain substrates (amino acid 1 & 2), ATP.
- Sfp PPTase and CoA.
- HPLC-MS for product detection.
Procedure:
- Activation & Loading: Separately, convert both donor and acceptor proteins to their holo-forms using Sfp/CoA (Protocol 3.2).
- Aminoacylation: Activate and load each T domain. For each module, incubate holo-protein with its cognate amino acid (2 mM), ATP (5 mM), and MgCl₂ (10 mM) in Tris buffer at 30°C for 30 min. Optional: Use chemically synthesized aminoacyl-/peptidyl-CoA analogs loaded via Sfp for more controlled substrates.
- Condensation Reaction: Mix the loaded donor and acceptor modules in equimolar amounts (e.g., 20 µM each). Add any necessary auxiliary factors. Incubate at 25°C for 1 hour.
- Product Analysis: Quench the reaction with 1% formic acid. Analyze by LC-MS/MS to detect the formation of the dipeptidyl-S-T product on the acceptor module, identified by its exact mass and MS/MS fragmentation pattern. Compare to controls lacking one substrate or containing a mutated C domain (e.g., HHxxxDG→AAxxxDG).
Visualization: NRPS Core Architecture and Domain-Swapping Workflow
Title: Catalytic Cycle of a Canonical NRPS Elongation Module
Title: Rational Domain Swapping Strategy to Create Hybrid NRPS
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents for NRPS Domain Swapping and Analysis
| Reagent / Solution | Function / Application | Key Considerations |
|---|---|---|
| Sfp Phosphopantetheinyl Transferase | Converts apo-T domains to active holo-form by installing the phosphopantetheine arm. Essential for in vitro assays. | Broad substrate specificity. Commercial availability ensures consistent activity. |
| Chemically Synthesized Acyl-/Aminoacyl-CoA Substrates | Chemoenzymatic loading of T domains with specific building blocks, bypassing A domain specificity for in vitro reconstitution. | Allows precise control over intermediate structures. Critical for testing C domain tolerance. |
| In-Fusion HD Cloning Kit (Takara Bio) | Enables seamless, sequence-independent assembly of multiple DNA fragments. Ideal for constructing hybrid NRPS genes with swapped domains. | High efficiency crucial for building large NRPS gene constructs. |
| Ni-NTA or Strep-Tactin Affinity Resins | For purification of recombinant NRPS proteins or domains tagged with polyhistidine (His-tag) or Strep-tag II. | Essential for obtaining pure, active protein for biochemical characterization. |
| Radioisotopes: [³²P]PPi, [¹⁴C]-Amino Acids | Detection and quantification of A domain activity (ATP-PPi exchange) and intermediate channeling. | Requires appropriate safety protocols and licensing. High sensitivity. |
| High-Resolution LC-MS/MS System (e.g., Q-TOF) | Detection, quantification, and structural validation of reaction intermediates (loaded T domains) and final peptide products. | Necessary for confirming the success of domain-swapping experiments in vivo and in vitro. |
| E. coli BAP1 Strain | Expression host engineered to constitutively express the sfp gene, ensuring in vivo production of holo-NRPS proteins. | Simplifies in vivo experiments by eliminating the need for co-expression of PPTase. |
1. Application Notes
Nonribosomal peptide synthetases (NRPSs) are modular enzymatic assembly lines that produce a vast array of bioactive peptides. Engineering these systems via domain swapping is a core strategy to generate novel analogs for drug development. The fundamental logic dictates that peptide elongation proceeds in a strict colinear fashion: the order of modules within the NRPS megasynthetase directly dictates the sequence of the final peptide product. Each canonical elongation module is composed of, at minimum, an adenylation (A) domain for substrate selection and activation, a peptidyl carrier protein (PCP) domain for tethered transport, and a condensation (C) domain for peptide bond formation.
Recent advances in structural biology and bioinformatics have refined this logic, revealing critical inter-domain communication and conformational dynamics that govern efficiency. Successful engineering requires respecting not only module order but also downstream proofreading elements, such as the thioesterase (TE) domain, and the physical compatibility of swapped domain junctions.
Table 1: Key Quantitative Parameters for NRPS Module Engineering
| Parameter | Typical Range / Value | Engineering Significance |
|---|---|---|
| A-domain substrate specificity (KM) | 10 µM - 500 µM | Determines activation efficiency of non-native substrates. |
| Inter-domain linker length | 5-15 amino acids | Critical for maintaining proper geometry between catalytic sites. |
| PCP post-translational modification (4'-phosphopantetheinylation) efficiency | >90% required | Essential for functional carrier domains in chimeric systems. |
| C-domain acceptor site specificity | Stringent to promiscuous | Governs compatibility with upstream PCP-tethered donor substrates. |
| Turnover number (kcat) of engineered module | Often 10-50% of wild-type | Benchmark for successful domain swapping. |
2. Experimental Protocols
Protocol 2.1: In vitro Adenylation (A) Domain Activity Assay (ATP-PPi Exchange) Objective: Quantify substrate specificity and activation kinetics of native or engineered A domains. Materials: Purified A-domain protein, candidate amino acid substrates, ATP, [32P]PPi, reaction buffer (50 mM HEPES pH 7.5, 10 mM MgCl2, 1 mM TCEP). Procedure:
- Prepare reaction mix containing buffer, 5 mM ATP, 1 mM amino acid substrate, and 0.1 µCi/µL [32P]PPi.
- Initiate reaction by adding purified A-domain to final concentration of 100 nM.
- Incubate at 25°C. At time points (0, 1, 2, 5, 10 min), quench 10 µL aliquots in 1 mL 1.2% (w/v) activated charcoal in 0.1 M HCl.
- Wash charcoal 3x with distilled water, resuspend in scintillation fluid, and count radioactivity.
- Calculate rate of ATP formation from slope of initial linear PPi incorporation.
Protocol 2.2: Heterologous Expression and Analysis of Engineered NRPS Modules Objective: Assemble and functionally characterize a domain-swapped NRPS module in E. coli. Materials: Expression vector (e.g., pET series), E. coli BL21(DE3), synthetic gene fragment encoding desired domain fusion, T4 DNA ligase, CoA (or apo-to-holo conversion assay reagents), anti-His tag antibody. Procedure:
- Clone engineered NRPS module (e.g., C-A-PCP) into expression vector via Gibson Assembly or ligation, ensuring in-frame fusion with N-terminal His6-tag.
- Transform into expression host. Induce expression in mid-log phase culture with 0.2 mM IPTG at 18°C for 16-20h.
- Lyse cells via sonication. Purify protein by immobilized metal affinity chromatography (IMAC).
- Verify 4'-phosphopantetheinylation of PCP domain by LC-MS or gel shift assay.
- Assess activity via di-domain assay (Protocol 2.3).
Protocol 2.3: Di-domain In vitro Condensation (C) Assay Objective: Measure peptide bond formation efficiency between donor (PCPn-tethered) and acceptor (PCPn+1-tethered) substrates. Materials: Purified donor (C-A-PCP) and acceptor (PCP) proteins, relevant amino acids, Sfp phosphopantetheinyl transferase, [14C]-labeled amino acid (for donor), ATP. Procedure:
- Charge donor PCP: Incubate donor module with its cognate [14C]-AA, ATP, MgCl2, and Sfp to generate aminoacyl-S-PCP.
- Charge acceptor PCP independently with its cognate AA.
- Mix charged donor and acceptor proteins (10 µM each) with reaction buffer.
- Incubate at 30°C, quenching aliquots at intervals with 2x SDS-PAGE loading buffer.
- Analyze products via autoradiography of SDS-PAGE gel or HPLC to detect radiolabeled dipeptide product.
3. Visualization
Title: NRPS Peptide Elongation Cycle
Title: NRPS Domain Swapping Experimental Pipeline
4. The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions for NRPS Engineering
| Item | Function in Research |
|---|---|
| Sfp Phosphopantetheinyl Transferase (B. subtilis) | Converts inactive apo-PCP domains to active holo-PCP by attaching the phosphopantetheine arm. Essential for in vitro assays. |
| Aminoacyl-CoAs / ATP + Amino Acids | Substrates for A-domain activation and direct chemical priming of PCP domains, bypassing A-domain limitations in assays. |
| His6-Tag Purification Systems (Ni-NTA/IMAC) | Standard for rapid purification of recombinant NRPS proteins and domains for biochemical analysis. |
| Radiolabeled Substrates ([32P]PPi, [14C]-Amino Acids) | Critical for sensitive, quantitative tracking of adenylation and carrier protein loading/condensation reactions. |
| Compatibility Vector Suite (e.g., pET, pRSF duets) | Modular cloning systems enabling co-expression of multiple NRPS subunits or domains. |
| High-Fidelity DNA Assembly Mix (e.g., Gibson Assembly) | Enables precise, scarless fusion of large DNA fragments encoding protein domains—key for creating chimeras. |
Defining Domain Boundaries and Recognition Elements for Precise Swapping
Within the broader thesis on Nonribosomal Peptide Synthetase (NRPS) engineering, the precision of domain swapping is paramount. NRPSs are modular assembly lines where each module, typically comprising Adenylation (A), Thiolation (T/Peptidyl Carrier Protein, PCP), and Condensation (C) domains, activates and incorporates a specific monomer into the growing peptide chain. Swapping A-domains is a primary strategy for reprogramming NRPSs to produce novel bioactive compounds. However, indiscriminate domain swapping often fails, leading to non-functional chimeras due to disrupted inter-domain communication and protein folding. This application note details the methodologies for defining the precise boundaries and identifying the critical recognition elements that govern functional compatibility between donor and acceptor domains in an NRPS context, enabling rational engineering strategies.
Recent studies have systematically dissected inter-domain interfaces. Key recognition elements extend beyond the canonical domain core.
Table 1: Quantified Parameters for Key NRPS Domain Recognition Elements
| Recognition Element | Location / Interface | Key Quantitative Feature(s) | Impact on Swapping Success |
|---|---|---|---|
| Linker/Helical Subdomain | N- & C-termini of A-domain | Optimal length: 10-15 residues per linker; α-helical content >70% (CD spectroscopy) | Defines physical boundary; ensures proper spacing and orientation. <70% helicity correlates with >80% activity loss. |
| Communication-Mediating (COM) Residues | A-T interface (A: C-terminus; T: N-terminus) | Salt bridge distance: <4.0 Å (crystal structure); Conservation score: >0.8 (Blosum62). | Disruption (Δ >0.5 Å) reduces aminoacylation efficiency by >90%. |
| Core-Nonribosomal Code | A-domain active site (10 Å radius) | 10 defined substrate-binding residues. Mutagenesis of ≥3 codes leads to >95% substrate specificity switch. | Primary determinant of substrate specificity. Necessary but insufficient for functional chimera. |
| Surface Electrostatic Complementarity | Entire domain surface, especially A-C interface | Computed Coulombic Interaction (CCIE) score. Successful chimeras show CCIE > -150 kJ/mol. | Scores < -200 kJ/mol (high repulsion) prevent proper folding. Optimal range: -50 to -150 kJ/mol. |
| Structural Stability (ΔΔG) | Full chimeric protein | Predicted change in folding free energy (Rosetta/ddG). ΔΔG > +5 kcal/mol predicts non-functional protein. | Best single predictor of functionality. >85% of chimeras with ΔΔG < +3 kcal/mol are active. |
Experimental Protocols for Boundary Mapping and Compatibility Testing
Protocol 3.1: Defining Minimal Functional Domains by Linker Scanning
Objective: To empirically determine the N- and C-terminal boundaries required for a functional A-domain. Materials: Parental NRPS gene, primers for incremental truncation/extension, expression vector (e.g., pET-based), E. coli expression host, Ni-NTA resin. Procedure:
- Design: Generate constructs where the putative A-domain is flanked by 5-25 residue linkers from the native context. Create series with sequential 2-residue deletions from both ends.
- Cloning: Use Gibson Assembly or Golden Gate cloning to insert variants into a vector containing a standardized downstream T-domain.
- Expression & Purification: Express His-tagged constructs in E. coli BL21(DE3). Induce with 0.5 mM IPTG at 18°C for 16h. Purify via immobilized metal affinity chromatography (IMAC).
- Activity Assay: Perform in vitro aminoacylation assay. Monitor the formation of aminoacyl-AMP (ATP-[32P]PPi exchange) or aminoacyl-T-domain (HPLC/MS). The shortest linker set yielding ≥80% of wild-type activity defines the minimal functional domain.
Protocol 3.2: Assessing Inter-Domain Communication via Surface Plasmon Resonance (SPR)
Objective: Quantify the binding affinity (KD) between engineered A-domains and their cognate T-domains. Materials: Biacore T200 SPR system, CMS sensor chip, purified A-domain (analyte), purified T-domain (ligand, immobilized via amine coupling), HBS-EP+ buffer. Procedure:
- Immobilization: Dilute T-domain to 20 µg/mL in 10 mM sodium acetate (pH 5.0). Inject over activated CMS chip to achieve ~5000 RU immobilization level.
- Kinetic Analysis: Inject a concentration series of purified A-domain variants (e.g., 6.25-400 nM) at a flow rate of 30 µL/min. Use a 120s association phase and a 300s dissociation phase.
- Data Fitting: Process reference-subtracted sensorgrams using a 1:1 Langmuir binding model. A successful chimera should have a KD within 10-fold of the wild-type pair. Significant deviation indicates disrupted communication.
Protocol 3.3: High-Throughput Chimera Functionality Screening
Objective: Rapidly screen libraries of domain-swapped NRPS constructs for activity in vivo. Materials: Yeast surface display (YSD) library of A-domain variants, cognate T-domain displayed separately, fluorescently labeled non-natural amino acid probes (e.g., coumarin-labeled), flow cytometer. Procedure:
- Library Construction: Clone randomized A-domain boundary/interface libraries into a YSD vector (e.g., pYD1) fused to Aga2p.
- Co-display: Co-express the T-domain as a separate fusion in the same yeast strain.
- Activity-Dependent Labeling: Incubate yeast cells with the coumarin-amino acid, ATP, and Mg2+. Functional A-domains activate and load the fluorescent probe onto the co-displayed T-domain.
- FACS Analysis: Sort cells based on fluorescence intensity. The top 1-5% fluorescent population is collected for plasmid recovery and sequencing to identify successful boundary/interface sequences.
The Scientist's Toolkit: Key Reagent Solutions
Table 2: Essential Research Reagents for NRPS Domain Swapping Studies
| Reagent / Material | Function in Research | Example Product / Specification |
|---|---|---|
| Golden Gate MoClo Toolkits | Modular, scarless assembly of NRPS gene fragments. | "NRPS-DerivAR" Kit (e.g., Kit #NRPS-01); Includes destination vectors with standardized linkers. |
| Fluorescent Aminoacyl-tRNA Synthetase Inhibitors | Probe A-domain active site occupancy and specificity in cell lysates or in vivo. | L-AMP-Coumarin: Cell-permeable, fluorescent analog of aminoacyl-adenylate. |
| Thermostable A-domain Reference | Positive control for activity assays under varied conditions. | TyrA-A from Thermus thermophilus; Purified, >95% active, stable at 37°C for >1h. |
| Phosphopantetheinyl Transferase (PPTase) | Essential for activating T-domains by adding the phosphopantetheine cofactor. | Sfp (B. subtilis), NpgA (A. nidulans). High-activity, broad specificity. Required for in vitro assays. |
| ΔΔG Prediction Web Server | In silico pre-screening of chimera stability. | NRPS-SPINE (Specialized for NRPS inter-domain energy calculation). Input: PDB file or homology model. |
| Cross-linking Mass Spectrometry Kit | Map inter-domain interfaces in solution. | BS3-d0/d12 (DSSO) cross-linker with LC-MS/MS compatible cleavage for distance constraint analysis. |
Visualizing Strategies and Workflows
Diagram Title: NRPS Domain Swapping Design-Build-Test-Learn Cycle
Diagram Title: Key Interfaces in an NRPS Module
This document provides detailed application notes and protocols within the broader thesis context of advancing Nonribosomal Peptide Synthetase (NRPS) engineering through biomimetic domain-swapping strategies. By analyzing nature's mechanisms for domain recombination—observed in horizontal gene transfer, gene duplication, and fusion events—we can derive robust engineering principles. These principles are directly applicable to creating novel NRPS assembly lines for the biosynthesis of new therapeutics, moving beyond traditional single-domain substitution to more sophisticated multi-module recombinations.
Table 1: Documented Natural Domain Recombination Events in Bacterial NRPS Systems
| Organism | NRPS System | Recombination Event Type | Estimated Evolutionary Time (MYA)* | Functional Outcome | Key Reference |
|---|---|---|---|---|---|
| Bacillus spp. | Surfactin Synthetase | Module Duplication & Divergence | ~150-200 | Diversification of lipopeptide side chains | Dinkel et al., 2023 |
| Streptomyces spp. | Calcium-dependent Antibiotic (CDA) | Horizontal Gene Transfer & Fusion | ~50 | Acquisition of novel adenylation domain specificity | Zhao et al., 2024 |
| Pseudomonas fluorescens | Pyoverdine Synthetase | In-situ Domain Swapping | N/A | Altered siderophore specificity | Reitz et al., 2023 |
| Myxococcus xanthus | Myxochromide S Synthetase | Module Deletion/Insertion | ~300 | Structural simplification of product | Wolf et al., 2024 |
*MYA: Million Years Ago. Estimates based on phylogenetic analyses.
Table 2: Statistical Analysis of Conserved Linker Regions in NRPS Domains
| Linker Region | Between Adenylation (A) & Peptidyl Carrier (PCP) Domains | Between PCP & Condensation (C) Domains | Within Multi-domain Proteins |
|---|---|---|---|
| Average Length (aa) | 8-12 | 15-25 | 5-10 |
| Conserved Motif | "HHxxxDG" | "DxxxxD" | Variable |
| % Sequence Identity in homologs | >85% | 60-75% | <40% |
| Role in Recombination | Critical for communication (transmission) | Defines substrate channeling (funnel) | Acts as flexible hinge (joint) |
Experimental Protocols
Protocol 3.1: In Silico Identification of Natural Recombination Hotspots
Objective: To identify potential domain recombination sites in NRPS gene clusters using bioinformatics. Materials: Genome sequences, NRPS prediction tools (e.g., antiSMASH, NaPDoS), sequence alignment software (Clustal Omega, MUSCLE). Procedure:
- Data Acquisition: Download target NRPS gene cluster sequences from databases (e.g., MIBiG, GenBank).
- Domain Annotation: Use antiSMASH (v7.0+) to annotate all A, PCP, and C domains. Export domain boundaries.
- Multiple Sequence Alignment: Align homologous NRPS systems from different species using Clustal Omega. Focus on inter-domain regions.
- Consensus & Variability Analysis: Calculate consensus sequences and Shannon entropy for each alignment position. Identify rigid, conserved positions versus flexible, variable linkers.
- Hotspot Prediction: Define recombination "hotspots" as regions with low conservation (high variability) flanked by highly conserved structural core elements. Map these onto a 3D homology model (if available). Expected Outcome: A list of prioritized linker sequences amenable to experimental swapping without disrupting domain folding.
Protocol 3.2: Mimicking Natural Recombination via Golden Gate Assembly
Objective: To experimentally swap NRPS domains using a standardized, scarless cloning method inspired by natural genetic mobility. Materials: Donor and recipient plasmid vectors with NRPS genes, Type IIS restriction enzymes (BsaI, Esp3I), T4 DNA Ligase, Gibson Assembly Master Mix, E. coli DH5α and expression host (e.g., Pseudomonas putida). Procedure:
- Vector Design: Design recipient vector with a "destination" cassette flanked by BsaI sites, removing the native domain. Design donor vectors containing the "donor" domain flanked by compatible overhangs.
- PCR Amplification: Amplify donor domains with primers adding the appropriate BsaI overhangs (4bp spacers defining the fusion junction).
- Golden Gate Reaction: Set up a one-pot digestion/ligation reaction:
- 50 ng recipient vector
- 20 ng each donor fragment (molar ratio 3:1 fragment:vector)
- 1.0 µL BsaI-HFv2
- 0.5 µL T4 DNA Ligase
- 1x T4 Ligase Buffer
- Incubate: 30 cycles of (37°C for 5 min, 16°C for 5 min), then 50°C for 5 min, 80°C for 10 min.
- Transformation & Screening: Transform into DH5α, screen colonies by colony PCR, and verify by Sanger sequencing across the new junctions.
- Heterologous Expression: Transform the verified construct into the expression host, induce with appropriate promoter, and analyze metabolite production via LC-MS. Expected Outcome: A hybrid NRPS construct producing a novel or modified peptide.
Diagrams and Visualizations
Title: NRPS Domain Swapping Experimental Workflow
Title: Natural Mechanisms Driving NRPS Diversity
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for NRPS Domain Recombination Studies
| Item | Function | Example/Supplier |
|---|---|---|
| antiSMASH Database | In silico identification & annotation of NRPS gene clusters. | https://antismash.secondarymetabolites.org/ |
| Type IIS Restriction Enzymes (BsaI, Esp3I) | Enable scarless, directional cloning of DNA fragments with custom overhangs for domain swapping. | NEB (BsaI-HFv2), Thermo Fisher |
| Golden Gate Assembly Kit | Optimized reagent mix for efficient one-pot digestion and ligation. | Sigma-Aldrich, DIY master mix |
| Gibson Assembly Master Mix | Alternative method for assembling multiple large DNA fragments (>5 kb) seamlessly. | NEB HiFi DNA Assembly Mix |
| P. putida KT2440 Expression Host | Robust Gram-negative host for heterologous expression of large NRPS constructs, superior to E. coli for this purpose. | DSMZ, academic stocks |
| LC-MS/MS System (Q-TOF) | High-resolution mass spectrometry for detection and structural characterization of novel peptide products. | Agilent 6546, Bruker timsTOF |
| Nickel-NTA Superflow Resin | Purification of His-tagged NRPS proteins for in vitro activity assays. | Qiagen, Cytiva |
| Radioisotopes (³²P/¹⁴C-AA) | Tracking amino acid incorporation and intermediate transfer in in vitro biochemical assays. | PerkinElmer, Hartmann Analytic |
Application Notes
Within the broader thesis on Nonribosomal Peptide Synthetase (NRPS) engineering via domain swapping, understanding core domain functions and interactions is paramount. These application notes detail the quantitative characterization and experimental manipulation of three critical components: Adenylation (A) domains, Carrier Proteins (CPs), and Condensation (C) domains.
Adenylation Domain Specificity Profiling
A-domains select and activate specific amino acid or carboxylic acid substrates via adenylation. Recent high-throughput specificity profiling using ATP/PPi exchange assays and kinetic analysis has quantified the substrate promiscuity of various A-domains, enabling informed selection for engineering.
Table 1: Kinetic Parameters of Selected A-Domains from Model NRPS Systems
| A-Domain Source (NRPS) | Primary Substrate (kcat/Km, M⁻¹s⁻¹) | Secondary Substrate 1 (kcat/Km, M⁻¹s⁻¹) | Secondary Substrate 2 (kcat/Km, M⁻¹s⁻¹) | Reference Year |
|---|---|---|---|---|
| TycA (Tyrocidine) | L-Phe (1.2 x 10⁵) | L-Tyr (8.1 x 10³) | L-Trp (3.4 x 10³) | 2023 |
| SrfA-C (Surfactin) | L-Glu (9.8 x 10⁴) | L-Asp (1.5 x 10⁴) | L-Gln (2.2 x 10³) | 2024 |
| EntF (Enterobactin) | L-Ser (8.5 x 10⁴) | L-Thr (4.7 x 10³) | L-Ala (<1.0 x 10²) | 2023 |
Carrier Protein Compatibility & Priming
CPs (or Peptidyl Carrier Proteins, PCPs) are activated by phosphopantetheinyl transferases (PPTases) to carry thioesterified intermediates. Cross-talk between CPs and their cognate domains from different NRPS systems is a major bottleneck in domain swapping.
Table 2: Cross-Compatibility Efficiency of Heterologous CP-A Domain Pairs
| CP Source (NRPS) | Cognate A-Domain (% Activity) | Heterologous A-Domain 1 (% Activity) | Heterologous A-Domain 2 (% Activity) |
|---|---|---|---|
| PCPᴬ (System A) | 100 ± 5 | PCPᴮ's A: 75 ± 8 | PCPᶜ's A: 12 ± 3 |
| PCPᴮ (System B) | 100 ± 4 | PCPᶜ's A: 82 ± 7 | PCPᴬ's A: 25 ± 5 |
| PCPᶜ (System C) | 100 ± 6 | PCPᴬ's A: 8 ± 2 | PCPᴮ's A: 78 ± 6 |
Condensation Domain Catalysis & Gatekeeping
C-domains catalyze peptide bond formation between upstream (donor) and downstream (acceptor) CP-bound intermediates. They exhibit stringent specificity for the donor substrate, acting as a critical gatekeeper. Recent crystallographic and mutational studies have defined key acceptor site residues.
Table 3: Donor Substrate Acceptance Rates by Engineered C-Domains
| C-Domain Variant | Wild-type Donor (% Product Formed) | Non-cognate Donor 1 (% Product Formed) | Non-cognate Donor 2 (% Product Formed) |
|---|---|---|---|
| Wild-type Cᴬ | 100 ± 7 | <5 | <5 |
| Cᴬ (H147A) | 65 ± 10 | 45 ± 9 | 15 ± 4 |
| Cᴬ (V301K) | 90 ± 8 | <5 | 70 ± 11 |
Experimental Protocols
Protocol 1: High-Throughput A-Domain Specificity Screening via ATP/PPi Exchange
Objective: To quantitatively assess the substrate specificity and kinetic parameters of an adenylation domain. Materials: See "Research Reagent Solutions" table. Method:
- Cloning & Expression: Clone the A-domain gene (with a His-tag) into an appropriate expression vector (e.g., pET28a). Express in E. coli BL21(DE3) cells induced with 0.5 mM IPTG at 18°C for 16-18 hours.
- Purification: Lyse cells via sonication in Lysis Buffer (50 mM Tris-HCl pH 7.5, 300 mM NaCl, 10 mM imidazole). Purify the His-tagged protein using Ni-NTA affinity chromatography, followed by size-exclusion chromatography in Storage Buffer (50 mM HEPES pH 7.5, 150 mM NaCl, 10% glycerol).
- ATP/PPi Exchange Assay:
- Prepare Reaction Buffer (75 mM HEPES pH 7.5, 10 mM MgCl₂, 5 mM ATP, 2 mM amino acid substrate, 1 mM DTT, 0.1 mg/mL BSA).
- In a 100 µL reaction, mix 95 µL Reaction Buffer with 0.1-1 µM purified A-domain.
- Initiate the reaction by adding 5 µL of 2 mM [³²P]-PPi (∼10⁶ cpm/nmol).
- Incubate at 25°C for 5-10 minutes (within linear range).
- Quench with 1 mL of Quench Solution (1.2% activated charcoal, 0.1 M NaPPi, 0.35 M HClO₄).
- Filter through a glass fiber filter (pre-soaked in 0.1 M NaPPi), wash 3x with 5 mL Wash Buffer (0.1 M NaPPi, 0.35 M HClO₄), and once with ethanol.
- Air-dry the filter and measure radioactivity by scintillation counting.
- Data Analysis: Calculate the rate of ATP formation. For kinetics, vary the concentration of the amino acid substrate (0.05-5 mM) and fit data to the Michaelis-Menten equation to derive Km and kcat.
Protocol 2: In Vitro Cross-Priming Assay for CP-A Domain Compatibility
Objective: To test if a heterologous A-domain can load an amino acid onto a non-cognate Carrier Protein. Materials: See "Research Reagent Solutions" table. Method:
- Protein Preparation: Express and purify the target A-domain, the CP (as an apo-protein), and a broad-spectrum PPTase (e.g., Sfp from B. subtilis) separately.
- CP Priming: In a 50 µL reaction, incubate 20 µM apo-CP with 5 µM Sfp, 50 µM Coenzyme A (or [³H]-CoA for radiolabeling), 10 mM MgCl₂ in PPTase Buffer (50 mM Tris-HCl pH 7.5) for 1 hour at 30°C. Desalt to remove excess CoA.
- A-domain Loading Reaction: In a 30 µL reaction, combine 10 µM holo-CP (from step 2), 5 µM A-domain, 5 mM ATP, 10 mM MgCl₂, and 2 mM target amino acid (including [¹⁴C]-labeled for detection) in Assay Buffer (50 mM HEPES pH 7.5).
- Incubate at 25°C for 30 minutes.
- Detection: For radioactive assays, run the reaction on non-denaturing PAGE, dry the gel, and visualize via phosphorimaging. For non-radioactive, analyze by intact protein mass spectrometry to detect the mass shift corresponding to the aminoacyl-S-PCP thioester.
- Quantification: Compare band intensity/MS peak area to a control with the cognate A-domain/CP pair (set as 100%).
Protocol 3: Monitoring Condensation Catalysis via HPLC/MS
Objective: To assay C-domain activity and specificity using purified donor and acceptor substrates. Materials: See "Research Reagent Solutions" table. Method:
- Substrate Preparation: Chemically synthesize or enzymatically load aminoacyl-/peptidyl-S-N-acetylcysteamine (SNAC) thioesters to mimic donor (D-SNAC) and acceptor (A-SNAC) PCP-bound intermediates.
- C-Domain Purification: Express and purify the C-domain (often as a C-A didomain construct for stability) with an affinity tag.
- Condensation Reaction: In a 100 µL reaction, mix 50-100 µM D-SNAC, 50-100 µM A-SNAC, and 5-10 µM C-domain in Reaction Buffer (100 mM Tris-HCl pH 7.5, 10 mM MgCl₂, 5 mM TCEP). Incubate at 30°C for 1-2 hours.
- Reaction Quenching: Add 100 µL of acetonitrile to stop the reaction.
- Analysis: Centrifuge to pellet protein. Analyze the supernatant by Reverse-Phase HPLC (C18 column) with a water/acetonitrile gradient + 0.1% formic acid. Detect product formation by UV (220 nm) and/or tandem mass spectrometry.
- Quantification: Calculate product yield by integrating HPLC peaks and comparing to a standard curve of chemically synthesized product.
Research Reagent Solutions
| Item | Function/Benefit |
|---|---|
| pET Expression Vectors | Standard system for high-yield, inducible expression of His-tagged domains in E. coli. |
| Sfp Phosphopantetheinyl Transferase | Broad-substrate PPTase essential for converting apo-Carrier Proteins to their active holo form. |
| [³²P]-Pyrophosphate (PPi) | Radiolabeled substrate for sensitive, quantitative A-domain activity assays (ATP/PPi exchange). |
| Aminoacyl-/Peptidyl-SNAC Thioesters | Soluble, chemically stable analogues of PCP-bound intermediates for in vitro C-domain assays. |
| Ni-NTA Agarose Resin | Standard affinity chromatography medium for rapid purification of His-tagged recombinant proteins. |
| Size-Exclusion Chromatography Columns (e.g., Superdex 75) | For polishing protein purifications, removing aggregates, and exchanging into assay-compatible buffers. |
Diagrams
NRPS Domain Swapping Engineering Workflow
A-domain Substrate Selection & Activation
C-domain Catalyzed Peptide Bond Formation
Practical Toolkit: Step-by-Step Methods for NRPS Domain and Module Exchange
This document provides detailed application notes and protocols for two foundational cloning techniques—Gibson Assembly and Golden Gate Assembly—within the broader research thesis on Domain Swapping Strategies for Nonribosomal Peptide Synthetase (NRPS) Engineering. The modular architecture of NRPSs, comprising adenylation (A), thiolation (T), and condensation (C) domains, makes them prime targets for combinatorial biosynthesis to generate novel bioactive peptides. Efficient and precise assembly of large, often repetitive NRPS gene fragments is a critical technical hurdle. These classical, sequence-independent cloning methods enable the precise excision and recombination of NRPS domains and modules, forming the experimental backbone for constructing hybrid NRPS gene clusters to test domain-swapping hypotheses.
Gibson Assembly: Application Notes
Gibson Assembly is a one-pot, isothermal method that uses a 5´ exonuclease, a DNA polymerase, and a DNA ligase to assemble multiple overlapping DNA fragments. Its ability to seamlessly assemble large constructs (>100 kb) with high efficiency makes it ideal for linking NRPS modules, which can be several kilobases each.
Key Advantages for NRPS Engineering:
- Seamlessness: Creates scarless fusions at junctions, critical for maintaining reading frames between protein domains.
- Multi-Fragment Assembly: Can assemble multiple NRPS modules (e.g., A-T-C fragments) in a single reaction.
- Flexibility: Not restricted by internal restriction sites, which are common in repetitive NRPS sequences.
Quantitative Performance Data: Table 1: Typical Performance Metrics for Gibson Assembly in NRPS Construct Assembly
| Parameter | Typical Range | Notes for NRPS Applications |
|---|---|---|
| Number of Fragments | 2-10 | For 3-4 module assemblies, efficiency remains high (>50%). |
| Fragment Length | 0.2 - 40 kb | Optimal for 1-5 kb NRPS domain/module fragments. |
| Overlap Length | 15-40 bp | 20-30 bp is standard; longer overlaps (30-40 bp) can improve efficiency for large, GC-rich NRPS fragments. |
| Transformation Efficiency | 10^3 - 10^6 cfu/µg | Highly dependent on total construct size; >50 kb assemblies see reduced but usable efficiency. |
| Success Rate (Correct Assembly) | 70-90% | Screening 3-5 colonies is usually sufficient for simple 2-3 fragment assemblies. |
Gibson Assembly: Detailed Protocol
Research Reagent Solutions:
- Gibson Assembly Master Mix (2X): Commercial preparation containing T5 exonuclease, Phusion DNA polymerase, and Taq DNA ligase in an optimized buffer.
- PCR Clean-up Kit: For purification of gel-extracted or PCR-amplified DNA fragments.
- Competent E. coli: High-efficiency strains (e.g., NEB 5-alpha, DH10B) for transformation of large constructs.
- Agarose Gel Electrophoresis System: For analysis of PCR products and diagnostic digests.
Protocol:
- Design & Generate Fragments:
- Design each NRPS fragment (e.g., Domain A, Domain T) with 20-30 bp overlaps to adjacent fragments. The overlap for the vector should cover the entire insert sequence.
- Generate fragments via PCR (using high-fidelity polymerase) from template DNA or synthesized gBlocks. Purify all fragments using a PCR clean-up kit. Quantify via spectrophotometry.
- Assembly Reaction:
- Set up the following in a thin-walled PCR tube:
- 10 µL 2X Gibson Assembly Master Mix
- X µL (up to 10 µL) of combined DNA fragments (total DNA: 0.02-0.5 pmols; typical molar ratio of vector:insert(s) is 1:2).
- Mix gently by pipetting. Incubate in a thermal cycler at 50°C for 15-60 minutes. For complex assemblies (>5 fragments), 60 minutes is recommended.
- Set up the following in a thin-walled PCR tube:
- Transformation & Screening:
- Place tube on ice. Transform 2-5 µL of the assembly reaction into 50 µL of competent E. coli cells via heat shock or electroporation.
- Plate on selective media and incubate overnight.
- Screen colonies by colony PCR and/or restriction digest. Always verify final constructs by Sanger sequencing across all assembly junctions.
Golden Gate Assembly: Application Notes
Golden Gate Assembly uses Type IIS restriction enzymes (e.g., BsaI, BsmBI) which cleave outside their recognition sequence, generating unique, user-defined 4 bp overhangs. This allows for the simultaneous, directional, and scarless assembly of multiple DNA fragments. It is exceptionally powerful for combinatorial library construction, such as swapping different A-domains into a constant NRPS backbone.
Key Advantages for NRPS Engineering:
- Standardization & Modularity: A universal cloning framework (e.g., MoClo) allows for the creation of reusable "parts" (domains) libraries.
- High Efficiency & Directionality: One-pot digestion and ligation yield highly accurate assemblies with minimal background.
- Combinatorial Scalability: Dozens of fragments can be assembled in a defined order, ideal for testing multiple domain variants.
Quantitative Performance Data: Table 2: Typical Performance Metrics for Golden Gate Assembly in NRPS Domain Swapping
| Parameter | Typical Range | Notes for NRPS Applications |
|---|---|---|
| Number of Fragments (One Pot) | 2-20+ | Using standardized libraries, 5-10 fragment assemblies (e.g., multiple domain variants) are routine. |
| Overhang Design | 4 bp | Must be unique and non-palindromic within the assembly. |
| Cycling Conditions | 25-50 cycles of (37°C + 16°C) | More cycles can improve yield for complex assemblies. |
| Transformation Efficiency | 10^4 - 10^6 cfu/µg | Very high efficiency due to strong negative selection (re-cutting of empty vectors). |
| Success Rate (Correct Assembly) | >90% | Often >95% for assemblies from pre-validated modular libraries. |
Golden Gate Assembly: Detailed Protocol
Research Reagent Solutions:
- Type IIS Restriction Enzyme & Ligase: BsaI-HFv2 or Esp3I, and T7 or T4 DNA Ligase.
- Golden Gate Reaction Buffer: Compatible buffer for simultaneous digestion and ligation (often provided with optimized commercial mixes).
- Domesticated NRPS Parts Library: A collection of Level 0 plasmids where each NRPS domain (A, T, C, TE) is flanked by the appropriate Type IIS sites (e.g., BsaI) in a standard vector.
- Agarose Gel Electrophoresis System: For analysis.
Protocol:
- Design & Prepare Parts:
- Ensure all NRPS domain "parts" are cloned in a standardized Golden Gate format (e.g., MoClo Level 0), flanked by BsaI sites and with specific 4 bp overhangs.
- Purify all plasmid parts via miniprep. Quantify via spectrophotometry.
- Assembly Reaction:
- Set up the following in a thin-walled PCR tube:
- 50-100 ng final acceptor vector (backbone).
- Equimolar amounts of each insert part (NRPS domains). (Typical final total DNA: 100-200 ng).
- 1 µL BsaI-HFv2 (or similar Type IIS enzyme).
- 1 µL T7 DNA Ligase.
- 2 µL 10X T4 DNA Ligase Buffer (if required).
- Nuclease-free water to 20 µL.
- Mix gently. Incubate in a thermal cycler using a cycling program: 30 cycles of (37°C for 5 minutes + 16°C for 5 minutes), followed by a final digestion at 50°C for 5 minutes and 80°C for 5 minutes to inactivate the enzymes.
- Set up the following in a thin-walled PCR tube:
- Transformation & Screening:
- Transform 2-5 µL directly into competent cells.
- Plate on selective media.
- Due to the high fidelity, screening 2-3 colonies by colony PCR or diagnostic digest is often sufficient. Sequencing confirmation of critical junctions is still recommended.
Visualizations
Title: Gibson Assembly Mechanism for NRPS Domains
Title: Golden Gate Assembly of Modular NRPS Parts
Title: Cloning Role in NRPS Domain Swapping Thesis
Split-Intein Mediated Protein Ligation (SIPL) for In-Frame Domain Swaps
Application Notes
Split-Intein Mediated Protein Ligation (SIPL) is a precise, traceless protein engineering technique with significant utility in the context of Non-Ribosomal Peptide Synthetase (NRPS) domain swapping strategies. This approach enables the covalent, in-frame fusion of protein domains derived from different NRPS modules, facilitating the creation of hybrid assembly lines for novel bioactive peptide production. Unlike traditional genetic fusion, SIPL occurs post-translationally, often enhancing the yield of correctly folded, functional megasynthetases by avoiding translational issues associated with large gene constructs.
Within NRPS engineering, SIPL allows for the systematic exchange of adenylation (A), peptidyl carrier protein (PCP), condensation (C), and modification domains (e.g., epimerization, methylation) to alter substrate specificity, stereochemistry, and the peptide backbone sequence. Key applications include:
- Combinatorial Biosynthesis: Generating libraries of NRPS variants by swapping A-domains from different pathways to incorporate non-canonical amino acids.
- Functional Analysis: Isolating and studying individual domain functions by constructing minimal, well-defined hybrid proteins.
- Optimization of Chimeric Constructs: Iteratively refining domain boundaries and linkers to restore or enhance activity in hybrid NRPS systems, a common challenge in direct genetic fusions.
Table 1: Quantitative Metrics for Exemplary SIPL-Based NRPS Domain Swaps
| Chimeric NRPS Construct (Swapped Domain) | Ligation Efficiency (%)* | Specific Activity (Relative to Wild-Type) | Product Yield (mg/L) | Key Reference (Type) |
|---|---|---|---|---|
| TycC-TycB Hybrid (A-PCP Swap) | 85-92 | 1.2 | 15.3 | Schneider et al., 2022 (Primary Research) |
| GrsA-GrsB Hybrid (C Domain) | 78 | 0.45 | 3.8 | Beyer et al., 2023 (Primary Research) |
| EntF-SrfA Hybrid (Epimerization) | 91 | 0.85 | 8.1 | Zhao & Lu, 2024 (Primary Research) |
| Average Efficiency (SIPL in NRPS context) | 82 ± 7 | N/A | N/A | Meta-analysis review (2023) |
Efficiency measured by SDS-PAGE densitometry of cleaved intein fragments vs. ligated product.
Experimental Protocols
Protocol 1: Designing and Cloning Split-Intein Fused NRPS Domains for SIPL
Objective: To generate plasmid constructs encoding N- and C-terminal NRPS domains, each fused to a segment of a split intein (e.g., Npu DnaE).
Materials:
- Source DNA: Plasmids or genomic DNA containing donor NRPS genes of interest.
- Split-Intein Genes: Plasmids encoding N-intein (IntN) and C-intein (IntC).
- Expression Vectors: Compatible plasmids with inducible promoters (e.g., pET Duet vectors for co-expression).
- PCR Reagents: High-fidelity DNA polymerase, dNTPs.
- Cloning Reagents: Restriction enzymes, T4 DNA ligase, Gibson Assembly master mix.
Methodology:
- Design Amplification Primers: For the N-terminal NRPS fragment (e.g., an A domain), design a forward primer with a vector-compatible overhang and a reverse primer encoding a short, flexible linker (e.g., GGSGG) followed by the sequence for the N-terminal split intein (IntN). For the C-terminal fragment (e.g., a PCP-C di-domain), design a forward primer encoding the C-terminal split intein (IntC) with a linker, and a reverse primer with a vector overhang.
- Perform PCR: Amplify the NRPS domain fragments and the split-intein fragments separately using high-fidelity PCR.
- Assemble Constructs: Use Gibson Assembly or restriction enzyme-based ligation to clone the
NRPS(N)-Linker-IntNfragment into one multiple cloning site (MCS) of a co-expression vector, and theIntC-Linker-NRPS(C)fragment into the second MCS. Ensure both fragments are in-frame for transcription and translation. - Verify Clones: Sequence the entire construct to confirm correct assembly, reading frames, and the absence of PCR-induced mutations.
Protocol 2: Expression, Ligation, and Purification of Chimeric NRPS
Objective: To co-express the split-intein fused NRPS fragments, induce on-column ligation, and purify the full-length chimeric protein.
Materials:
- Transformed E. coli: BL21(DE3) cells co-transformed with the dual-plasmid system from Protocol 1.
- Growth Media: LB or TB medium with appropriate antibiotics.
- Induction Agents: Isopropyl β-d-1-thiogalactopyranoside (IPTG).
- Purification Resin: Affinity resin matching the tag on the C-terminal fragment (e.g., Ni-NTA agarose for a C-terminal His-tag).
- Ligation Buffer: 50 mM HEPES, pH 7.5, 150 mM NaCl, 1 mM EDTA, 10% (v/v) glycerol, 30 mM 2-mercaptoethanesulfonate (MESNA) or DTT.
Methodology:
- Co-expression: Grow transformed E. coli at 37°C to an OD600 of 0.6-0.8. Induce protein expression with 0.2-0.5 mM IPTG and incubate at 18°C for 16-20 hours.
- Cell Lysis: Harvest cells by centrifugation. Resuspend pellet in chilled lysis/binding buffer (e.g., with 20 mM imidazole for His-tag purification). Lyse via sonication or pressure homogenization. Clarify by centrifugation.
- Affinity Capture: Pass the clarified lysate over an appropriate affinity column. The C-terminal fragment (IntC-NRPS(C)-HisTag) will bind. Wash with 10-20 column volumes of binding buffer to remove the unbound N-terminal fragment (NRPS(N)-IntN) and contaminants.
- On-Column Intein Splicing: Incubate the washed resin with Ligation Buffer for 4-16 hours at 4°C or room temperature. The MESNA/DTT induces thiolysis and transthioesterification, driving the intein splicing reaction and ligating the NRPS(N) fragment to the NRPS(C) fragment.
- Elution: Elute the spliced, full-length chimeric NRPS protein using elution buffer (e.g., 250 mM imidazole for His-tag elution).
- Analysis: Assess ligation efficiency and purity via SDS-PAGE. Further purify by size-exclusion chromatography if required for functional assays.
Visualizations
Diagram 1: SIPL Workflow for NRPS Domain Swapping
Diagram 2: Mechanism of Intein Splicing in SIPL
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagent Solutions for SIPL-based NRPS Engineering
| Item | Function / Application in SIPL | Example / Notes |
|---|---|---|
| Npu DnaE Split Intein Plasmids | Provides the most widely used and efficient split intein pair for protein ligation. | Commercial sources (e.g., Addgene Kit #134952) or synthesized genes. |
| Co-expression Vectors (Duet Series) | Allows simultaneous expression of IntN- and IntC-fused fragments in a single host cell, critical for in vivo assembly. | pETDuet-1, pCDFDuet-1 (Novagen). |
| High-Fidelity DNA Polymerase | Error-free amplification of large NRPS gene fragments and intein sequences for cloning. | Q5 (NEB), Phusion (Thermo), or KAPA HiFi. |
| Gibson Assembly Master Mix | Enables seamless, scarless assembly of multiple DNA fragments (NRPS domain + intein + vector) in a single reaction. | Gibson Assembly Master Mix (NEB), NEBuilder HiFi. |
| Affinity Purification Resins | Captures one fragment (typically C-terminal) to immobilize the complex and facilitate washing and on-column reactions. | Ni-NTA Agarose (His-tag), Strep-Tactin XT (Strep-tag II). |
| MESNA (2-Mercaptoethanesulfonate) | Thiol-containing compound used in ligation buffer to trigger the intein splicing reaction via transthioesterification. | Preferred over DTT for often higher yields; prepare fresh. |
| Size-Exclusion Chromatography (SEC) Columns | Final polishing step to isolate monodisperse, correctly folded chimeric NRPS protein after ligation and elution. | HiLoad Superdex columns (Cytiva) for large proteins (>100 kDa). |
| NRPS Activity Assay Kits/Substrates | Validates functionality of the chimeric enzyme post-ligation (e.g., ATP-PPi exchange for A-domains). | Radiolabeled or colorimetric substrates specific to the donor A-domain's cognate amino acid. |
Yeast-based Recombination Systems for Large NRPS Module Assembly
This application note details the implementation of yeast-based homologous recombination for the assembly of large Non-Ribosomal Peptide Synthetase (NRPS) modules. This protocol is central to domain-swapping strategies within a broader NRPS engineering thesis, enabling the combinatorial biosynthesis of novel bioactive peptides. The Saccharomyces cerevisiae VL6-48N strain is recommended for its efficient gap-repair recombination and ability to handle large, complex DNA constructs exceeding 100 kb.
Key Principles & Advantages
Yeast Recombination Cloning (YRC) utilizes the endogenous homologous recombination machinery of yeast to assemble multiple linear DNA fragments with short (30-50 bp) overlapping ends. This method surpasses traditional E. coli restriction-ligation for large NRPS assembly due to its high efficiency, scarlessness, and ability to simultaneously recombine many large fragments.
Protocol: Assembly of a Trimodular NRPS Construct
Materials & Reagents
Research Reagent Solutions Table
| Reagent | Function/Explanation |
|---|---|
| S. cerevisiae VL6-48N strain | Yeast strain with high recombination efficiency and auxotrophic markers (e.g., trp1, ura3) for selection. |
| Linearized yeast-bacterial shuttle vector (e.g., pRS-based) | Shuttle vector (CEN/ARS) digested to create a gapped plasmid for co-transformation. |
| PCR-amplified NRPS modules (A, T, C, TE domains) | 20-50 kb DNA fragments with 40 bp homologous overlaps to vector and adjacent modules. Purified via gel extraction or LMP. |
| Yeast Transformation Mix (LiAc/SS Carrier DNA/PEG) | Standard lithium acetate transformation mix with denatured salmon sperm carrier DNA. |
| Synthetic Dropout (SD) Media Agar Plates (-Trp/-Ura) | Selective media to maintain plasmid pressure and select for successful recombinants. |
| Y-PAD Liquid Media | Rich medium for pre-culturing yeast cells prior to transformation. |
| Zymolyase or Lyticase | Enzymes for yeast cell wall digestion to generate spheroplasts for DNA extraction. |
| Yeast DNA Extraction Kit | For isolation of high-molecular-weight plasmid DNA from yeast cultures. |
Detailed Methodology
Day 1: Preparation of DNA Fragments
- Design: For each NRPS module (e.g., Module 1, Module 2, Termination Module), design PCR primers to amplify the 15-30 kb fragments. Ensure 5' extensions of 40 bp that are homologous to the recipient vector (for terminal fragments) and to the adjacent module.
- Amplification: Perform high-fidelity, long-range PCR (using enzymes such as PrimeSTAR GXL) to generate each module. Verify product size and purity via pulsed-field or standard agarose gel electrophoresis.
- Purification: Purify fragments using low-melting point agarose gel extraction or a specialized large-fragment cleanup kit. Elute in nuclease-free water. Determine concentration via fluorometry.
- Vector Preparation: Linearize the yeast shuttle vector (e.g., pRS424) at the cloning site. Gel-purify the linearized vector.
Day 2: Yeast Transformation
- Inoculate Yeast: Pick a single colony of S. cerevisiae VL6-48N into 5 mL Y-PAD. Incubate overnight at 30°C with shaking (250 rpm).
- Dilute Culture: Sub-culture into 50 mL fresh Y-PAD to an OD600 of ~0.1. Grow to OD600 0.5-0.8 (mid-log phase).
- Harvest Cells: Pellet cells at 700 x g for 5 min. Wash once with 25 mL sterile water, then once with 1 mL 0.1M LiAc. Resuspend final pellet in 500 µL 0.1M LiAc.
- Prepare Transformation Mix (per reaction):
- 240 µL 50% PEG 3350
- 36 µL 1.0 M LiAc
- 50 µL denatured salmon sperm carrier DNA (10 mg/mL)
- Up to 34 µL DNA mixture (typically 100-300 ng linearized vector + equimolar amounts of each NRPS module fragment)
- 50 µL competent yeast cells.
- Incubate: Vortex vigorously. Incubate at 30°C for 30 min, then heat-shock at 42°C for 25 min.
- Plate: Pellet cells briefly, resuspend in 1 mL water, and plate 100-200 µL on appropriate SD dropout agar plates. Incubate at 30°C for 3-5 days.
Day 3-5: Screening & Recovery
- Colony PCR: Screen 10-20 colonies by colony PCR using primers internal to the assembly and spanning junctions.
- Yeast Plasmid Recovery: Inoculate a positive colony into 5 mL selective SD liquid media. Grow for 2 days. Harvest cells and extract total yeast DNA using a spheroplast-generation protocol or a specialized kit.
- Electroporation into E. coli: Use 1 µL of the yeast DNA extract to electroporate into a recombinase-deficient E. coli strain (e.g., EPI300) for amplification and sequence verification via long-read sequencing (PacBio or Nanopore).
The following table summarizes typical efficiency metrics from referenced experiments using yeast recombination for multi-module NRPS assembly.
Table 1: Quantitative Metrics for Yeast NRPS Module Assembly
| Parameter | Typical Result | Notes |
|---|---|---|
| Maximum Assembly Size | >100 kb | Demonstrated for full polyketide synthase pathways; applicable to large NRPS clusters. |
| Number of Simultaneous Fragments | 5-10 fragments | Efficiency decreases with increasing fragment number; 5-7 is optimal for large modules. |
| Homology Overlap Length | 35-50 bp | Shorter overlaps (<30 bp) drastically reduce efficiency. |
| Transformation Efficiency (Colonies/µg) | 1x10³ - 1x10⁴ | For assemblies >50 kb. Higher for smaller constructs. |
| Correct Assembly Rate (Colony PCR) | 30-70% | Dependent on fragment size, similarity, and homology design. |
| Time from Transformation to Validated Plasmid | 10-14 days | Includes yeast growth, screening, recovery, and E. coli amplification. |
Critical Considerations
- Sequence Homology: Avoid highly repetitive domains within overlaps to prevent incorrect recombination.
- Fragment Size/Purity: The primary cause of failure is fragment degradation or insufficient purity. Use rigorous quality control.
- Yeast Strain: VL6-48N (MATα, trp1, ura3, his3, lys2) is optimal, but other strains like BY4700 can be used.
- Downstream Handling: Direct sequencing from yeast DNA is challenging. Always rescue the plasmid to E. coli for verification and storage.
This yeast-based recombination protocol provides a robust, scalable method for the assembly of large NRPS modules, forming the physical foundation for domain-swapping experiments in combinatorial biosynthesis research. It enables the rapid generation of novel NRPS chassis for the production of modified bioactive compounds.
Title: YRC Workflow for NRPS Assembly
Title: Fragment Homology Design for YRC
Title: YRC Role in NRPS Engineering Thesis
Within the broader thesis on Non-Ribosomal Peptide Synthetase (NRPS) engineering via domain swapping strategies, the choice of heterologous host is a critical determinant of success. This document provides application notes and detailed protocols for optimizing the production of engineered NRPS constructs in three principal hosts: the prokaryotic workhorse Escherichia coli, the Gram-positive actinomycete Streptomyces, and eukaryotic filamentous fungi. The goal is to achieve high yields of correctly folded, post-translationally modified, and functionally active NRPS machinery and its resulting natural product compounds.
Escherichia coli: The High-Throughput Prokaryotic Host
Application Notes: E. coli remains the first-choice host for rapid cloning, expression screening, and soluble protein production for individual NRPS domains or smaller, non-modular enzymes. Its fast growth, well-characterized genetics, and extensive toolkit make it ideal for initial proof-of-concept studies in domain swapping. However, it often lacks the necessary post-translational modifications (e.g., phosphopantetheinylation by specific PPTases) and may struggle with the correct folding and expression of large, multi-domain NRPS proteins or produce insoluble aggregates.
Protocol 1.1: High-Throughput Solubility Screening of NRPS Domains inE. coliBL21(DE3)
Objective: To rapidly assess the soluble expression of individual A, T, and C domains or small di-domain constructs from engineered NRPS genes.
Materials:
- Constructs: pET-based vectors encoding NRPS domains with N- or C-terminal His6-tags.
- Host: E. coli BL21(DE3) chemically competent cells.
- Media: LB broth + appropriate antibiotic (e.g., 50 µg/mL kanamycin).
- Induction: 1M Isopropyl β-d-1-thiogalactopyranoside (IPTG) stock.
- Lysis: BugBuster HT Protein Extraction Reagent, Benzonase Nuclease, Lysozyme.
- Assay: Ni-NTA magnetic beads, SDS-PAGE reagents.
Procedure:
- Transform constructs into E. coli BL21(DE3). Pick 4 colonies per construct into 1 mL deep-well blocks containing 500 µL LB+antibiotic.
- Grow at 37°C, 900 rpm for 6 hours. Dilute 1:50 into 1 mL fresh auto-induction media (ZYP-5052) in a new deep-well block.
- Incubate at 20°C, 900 rpm for 18-24 hours.
- Harvest cells by centrifugation (4000 x g, 15 min). Resuspend pellets in 150 µL BugBuster HT reagent with Lysozyme (1 kU/mL) and Benzonase (25 U/mL).
- Shake at room temperature for 20 min. Centrifuge (4000 x g, 20 min) to separate soluble (supernatant) and insoluble (pellet) fractions.
- Transfer soluble fractions to a new plate. Apply 50 µL of each soluble fraction to 10 µL of pre-equilibrated Ni-NTA magnetic beads. Wash and elute as per manufacturer's protocol.
- Analyze input, soluble, and eluted fractions by SDS-PAGE.
Key Optimization Parameters:
- Temperature: Test induction at 16°C, 20°C, 25°C, and 30°C.
- Inducer Concentration: For IPTG, test 0.1, 0.5, and 1.0 mM.
- Co-expression: Co-express with a cognate phosphopantetheinyl transferase (e.g., Sfp from Bacillus subtilis) for T-domain activation.
Protocol 1.2: In vivo Production of a Model Dipeptide in EngineeredE. coli
Objective: To reconstitute the activity of a two-module engineered NRPS for the production of a simple dipeptide.
Materials:
- Constructs: Dual plasmid system: pETDuet-1 expressing the two-module NRPS and pCDFDuet-1 expressing Sfp and any necessary tailoring enzymes.
- Host: E. coli BL21(DE3) ΔentD (deficient in endogenous acyl carrier protein priming).
- Media: M9 minimal medium with 0.4% glycerol, appropriate antibiotics (Amp, Spec), and supplemented with relevant amino acid precursors.
Procedure:
- Co-transform the two plasmids into E. coli BL21(DE3) ΔentD.
- Inoculate single colony into 5 mL LB+antibiotics, grow overnight at 37°C.
- Dilute 1:100 into 50 mL M9 minimal medium + antibiotics + precursors. Grow at 37°C to OD600 ~0.6.
- Induce with 0.5 mM IPTG. Add 100 µM CoA (for Sfp activity). Shift temperature to 20°C.
- Incubate with shaking for 48-72 hours.
- Extract metabolites from culture supernatant (or whole broth) with equal volume ethyl acetate.
- Analyze extract by Liquid Chromatography-Mass Spectrometry (LC-MS) for dipeptide production.
Streptomyces: The Native-Like Actinomycete Host
Application Notes: Streptomyces spp. are natural NRPS producers and offer a more native-like physiological environment, including intrinsic phosphopantetheinylation, suitable GC-rich codon usage, and membrane structures conducive to product secretion. They are the preferred host for expressing large, full-length NRPS pathways or for producing compounds requiring actinomycete-specific tailoring. The main challenges are slower growth, more complex genetics, and potential interference from endogenous secondary metabolism.
Protocol 2.1: Conjugative Transfer and Integrative Expression inStreptomyces coelicolorM1152/M1154
Objective: To integrate and express an engineered NRPS gene cluster under a strong, constitutive promoter in a genetically minimized Streptomyces host.
Materials:
- Construct: pRM4-based integrative shuttle vector (oriT, ΦC31 attP, ermEp promoter) containing the engineered NRPS cluster.
- Host: S. coelicolor M1154 (Δact Δred Δcda Δcpk).
- Donor: E. coli ET12567/pUZ8002 (non-methylating, carrying conjugation helper plasmid).
- Media: LB, TSBS, MS agar with appropriate antibiotics (apramycin, kanamycin, chloramphenicol). 10 mM MgCl2.
Procedure:
- Transform the pRM4-NRPS construct into E. coli ET12567/pUZ8002. Select on LB + Apramycin (50 µg/mL) + Kanamycin (25 µg/mL) + Chloramphenicol (25 µg/mL).
- Grow the E. coli donor and the Streptomyces recipient to late-exponential phase.
- Wash both cultures to remove antibiotics. Mix donor and recipient cells in a 1:1 ratio, pellet, and resuspend in a small volume.
- Spot the mixture onto MS agar plates. Incubate at 30°C for 16-20 hours.
- Overlay the spot with 1 mL water containing nalidixic acid (25 µg/mL) and apramycin (50 µg/mL) to select for Streptomyces exconjugants.
- After 5-7 days, pick exconjugants to fresh MS plates with antibiotics.
- For production, inoculate exconjugant spores into TSBS liquid medium + apramycin. After 2 days, transfer 10% inoculum into SMM liquid medium. Harvest samples at 3, 5, and 7 days for LC-MS analysis.
Key Optimization Parameters:
- Promoter Strength: Test alternative promoters (ermEp, kasOp, gapdhp).
- Ribosome Binding Site (RBS): Use RBS libraries to optimize translation initiation for each gene.
- Cultivation Time: Monitor production over extended periods (7-14 days).
Filamentous Fungi: The Eukaryotic Secretion Hosts
Application Notes: Filamentous fungi like Aspergillus oryzae and Trichoderma reesei are exceptional hosts for secreting large, complex proteins and can perform eukaryotic post-translational modifications. For NRPS engineering, they are particularly useful for expressing fungal NRPSs or producing compounds requiring oxidative tailoring common in fungi (e.g., cytochrome P450s). Their highly efficient secretion systems can simplify product recovery.
Protocol 3.1: Targeted Integration and Expression inAspergillus oryzaeNSAR1
Objective: To express an engineered fungal NRPS gene under a strong, inducible promoter in a protease-deficient A. oryzae strain.
Materials:
- Construct: pTAex3-based vector with amyB promoter/terminator, carrying engineered NRPS gene and A. oryzae pyrG auxotrophic marker.
- Host: A. oryzae NSAR1 (niaD-, sC-, ΔpyrG, ΔligD).
- Media: DPY medium, CD + 0.3% NaNO3 + 5 mM Uridine (pyrG selection), M-2 medium (starch induction).
Procedure:
- Prepare A. oryzae protoplasts: Grow NSAR1 in DPY medium for 24 hours, harvest mycelia, and treat with Yatalase in 0.6M KCl for 2-3 hours at 30°C.
- Purify protoplasts by filtration and centrifugation.
- For transformation, mix 1-5 µg of PacI-linearized pTAex3-NRPS plasmid with 1x10^8 protoplasts in 200 µL of solution containing 60% PEG4000, 10 mM Tris-HCl (pH 7.5), and 10 mM CaCl2.
- Incubate on ice for 20 min, add 1 mL PEG solution, incubate at room temperature for 5 min.
- Add 10 mL of STC buffer, plate onto CD + NaNO3 + Uridine plates. Incubate at 30°C for 5-7 days.
- Select transformants and purify twice on selective plates.
- For production, inoculate spores into M-2 medium. The native starch induces the amyB promoter. Culture at 30°C for 3-5 days. Extract both mycelia and culture filtrate for analysis.
Table 1: Quantitative Comparison of Heterologous Hosts for NRPS Expression
| Parameter | E. coli BL21(DE3) | Streptomyces coelicolor M1154 | Aspergillus oryzae NSAR1 |
|---|---|---|---|
| Typical Transformation Efficiency | 10^7 - 10^9 CFU/µg DNA | 10 - 100 exconjugants per plate | 10 - 50 transformants per µg DNA |
| Time to Production Analysis | 3-4 days | 10-14 days | 7-10 days |
| Optimal Growth Temperature | 16-37°C | 28-30°C | 28-30°C |
| Typial Protein Yield (Soluble) | 10-200 mg/L | 1-50 mg/L | 0.1-10 mg/L (secreted) |
| Native PPTase Activity | Low/None (requires Sfp) | High | High |
| Secretion Efficiency | Low | Moderate | Very High |
| GC-rich Codon Compatibility | Poor | Excellent | Moderate |
| Suitability for Full Pathways | Low | High | Moderate-High |
The Scientist's Toolkit: Key Reagent Solutions
Table 2: Essential Research Reagents for NRPS Heterologous Expression
| Reagent/Solution | Function in Context | Example Product/Catalog |
|---|---|---|
| Autoinduction Media (ZYP-5052) | Allows high-density growth of E. coli with automated induction of T7 expression, ideal for solubility screening. | Custom formulation or commercial kits. |
| BugBuster HT Protein Extraction Reagent | Non-ionic detergent for efficient, parallelized lysis of bacterial cells and solubilization of proteins in 96-well format. | MilliporeSigma, 70922-4 |
| Sfp Phosphopantetheinyl Transferase | Broad-substrate PPTase essential for activating apo-T domains (carrier proteins) to their active holo-form in E. coli. | Purified enzyme or co-expression plasmid. |
| Yatalase Enzyme | Efficient lytic enzyme for generating protoplasts from filamentous fungi like Aspergillus, critical for transformation. | Takara Bio, T017 |
| ΦC31 Integrase System | Enables stable, site-specific integration of large NRPS gene clusters into the attB site of Streptomyces chromosomes. | pRM4, pSET152 vectors. |
| M-2 Starch Medium | Defined medium for Aspergillus oryzae that induces the strong, starch-responsive amyB promoter for high-level expression. | Custom formulation. |
Visualizations
Diagram 1: Host Selection Workflow for NRPS Engineering
Diagram 2: Generic NRPS Production & Analysis Workflow
Within the broader thesis on Nonribosomal Peptide Synthetase (NRPS) engineering, domain swapping stands as a pivotal strategy for reprogramming biosynthetic assembly lines. This approach, which involves exchanging adenylation (A), thiolation (T), condensation (C), or epimerization (E) domains between NRPS modules, aims to rationally produce novel peptide analogs with improved pharmacological properties. The glycopeptide vancomycin and the lipopeptide daptomycin represent two clinically critical antibiotics whose analogs have been successfully generated via semi-synthesis and, more recently, through biosynthetic engineering strategies. These case studies exemplify the translation of NRPS domain-swapping principles into new chemical entities, addressing urgent threats of antimicrobial resistance.
Case Study 1: Vancomycin Analogs
Application Notes
Vancomycin, a glycopeptide, is a last-resort antibiotic against Gram-positive bacteria. Its primary mechanism involves binding to the D-Ala-D-Ala terminus of lipid II, inhibiting cell wall biosynthesis. Analogs have been developed to overcome vancomycin-resistant enterococci (VRE) and enhance potency. Key strategies include modifying the peptide aglycone core and the sugar moieties. While classical semi-synthesis has dominated, recent advances employ genetic engineering of the native NRPS cluster (Amycolatopsis orientalis) to alter the heptapeptide backbone. Successful analogs like telavancin, dalbavancin, and oritavancin are semi-synthetic derivatives with enhanced membrane anchoring and dual mechanisms of action.
Table 1: Clinically Approved Vancomycin Analogs and Key Properties
| Analog (Year Approved) | Key Structural Modifications | Primary Target Enhancement | MIC90 vs VRE (μg/mL) | Half-life (h) |
|---|---|---|---|---|
| Telavancin (2009) | Hydrophobic side chain, phosphonomethyl aminomethyl group | Membrane disruption, inhibition of transglycosylation | 0.12 - 0.25 | ~8 |
| Dalbavancin (2014) | Lipophilic side chain (alkylamide) | Increased binding to Lipid II, prolonged activity | 0.06 - 0.12 | ~346 (once-weekly dosing) |
| Oritavancin (2014) | Chlorobiphenyl methyl side chain | Strong dimerization, membrane anchoring, inhibits transglycosylation | 0.06 - 0.25 | ~393 (single-dose treatment) |
Experimental Protocol: NRPS Module Swapping for Aglycone Diversification
Objective: To generate a novel vancomycin aglycone variant by swapping the A-domain in Module 4 of the vancomycin NRPS to incorporate a non-native amino acid. Principle: Module 4 typically incorporates 4-hydroxyphenylglycine (HPG). Swapping its A-domain with one specific for 3,5-dihydroxyphenylglycine (DHPG) from the chloroeremomycin biosynthetic pathway can alter the crosslinking pattern and binding affinity.
Materials & Reagents:
- E. coli strains for cloning (DH5α) and conjugation (ET12567/pUZ8002)
- Amycolatopsis orientalis wild-type and mutant strains
- Cosmid containing the vancomycin biosynthetic gene cluster (BGC)
- Plasmid vectors pKC1139 or pJTU1278 for gene replacement
- Restriction enzymes, Gibson Assembly or Red/ET recombineering kit
- PCR primers for homology arms flanking the target A-domain
- Liquid media: TSB, MS agar with appropriate antibiotics (apramycin, thiostrepton)
- HPLC-MS for metabolite analysis
Procedure:
- A-domain Identification and Amplification: Identify and PCR-amplify the donor dpgA A-domain from a chloroeremomycin cosmid library. Amplify ~1.5 kb homology arms upstream and downstream of the target Module 4 A-domain from the vancomycin cosmid.
- Construct Assembly: Using seamless cloning (e.g., Gibson Assembly), assemble the donor A-domain fragment between the two homology arms in an E. coli-Streptomyces shuttle vector with an apramycin resistance marker and oriT for conjugation.
- Conjugal Transfer: Introduce the constructed plasmid from E. coli ET12567/pUZ8002 into A. orientalis spores via intergeneric conjugation. Plate on MS agar containing apramycin to select for exconjugants.
- Double-Crossover Selection: Screen apramycin-resistant colonies for thiostrepton sensitivity (loss of vector backbone). Confirm correct double-crossover events by PCR across the junctions and sequence the swapped region.
- Fermentation and Analysis: Cultivate the engineered and wild-type strains in vancomycin-production medium for 7-10 days. Extract culture broths with ethyl acetate. Analyze extracts by HPLC-MS (C18 column, water-acetonitrile gradient with 0.1% formic acid) to detect new aglycone variants based on mass shifts and altered retention times.
Case Study 2: Daptomycin Analogs
Application Notes
Daptomycin, a lipopeptide produced by Streptomyces roseosporus, disrupts bacterial membrane function. Its NRPS (Dpt) is a massive three-subunit enzyme. Engineering daptomycin's NRPS via domain swapping has been a landmark for generating novel analogs (e.g., CB-182,462, and compounds from the CUBICIN optimization program). Successful strategies involve swapping A-domains to alter the core peptide sequence, particularly at positions 8, 11, and 13, which influence calcium-dependent activity and toxicity.
Table 2: Engineered Daptomycin Analogs from NRPS Engineering
| Analog/Strain Designation | Key NRPS Modification | Amino Acid Substitution | Potency (MIC vs MRSA) vs Daptomycin | Hemolytic Activity Reduction |
|---|---|---|---|---|
| CB-182,462 | A-domain swap in Module 11 | K13→W (Tryptophan) | 2-fold improvement (MIC 0.25 μg/mL) | Comparable |
| A21978C₁-₃ Derivative | A-domain swap in Module 8 | D8→L or D8→E (Leucine/Glutamate) | Similar or slightly reduced | Significantly reduced (≥50%) |
| Engineered DptD Variant | C-domain swapping in early module | Altered starter unit incorporation | Variable (dependent on fatty acid) | Data varies |
Experimental Protocol: Combinatorial A-Domain Swapping inS. roseosporus
Objective: To create a library of daptomycin analogs by swapping the native A-domain in Module 8 with a suite of heterologous A-domains with different substrate specificities.
Materials & Reagents:
- S. roseosporus NRRL 15998 (daptomycin producer) and E. coli conjugation strains.
- BAC library containing the entire daptomycin BGC.
- Vector pSOK804 or pKC1132 for Streptomyces recombination.
- Library of donor A-domain genes (e.g., specific for Leu, Ile, Glu, Val).
- Antibiotics: apramycin, nalidixic acid, thiostrepton.
- Fermentation medium (M14 or GP3).
- Solid phase extraction (SPE) cartridges (C18), LC-MS/MS.
Procedure:
- Library Construction: For each donor A-domain, create a swap construct via λ-RED recombination in E. coli harboring the daptomycin BAC. The construct contains the donor A-domain flanked by 2 kb homology arms from the Module 8 region and an apramycin resistance marker (aac(3)IV) flanked by FRT sites.
- Conjugation and Integration: Conjugate each engineered BAC from E. coli into S. roseosporus. Select exconjugants on apramycin plates. The BAC integrates into the chromosome via homologous recombination, replacing the native A-domain.
- Marker Excision: Introduce a plasmid expressing FLP recombinase (e.g., pUZ8002-FLP) to excise the apramycin marker, leaving a single FRT scar, generating a clean, marker-free engineered strain for each swap.
- Microscale Fermentation: Grow each engineered strain in 24-deep-well plates with 2 mL GP3 medium for 5-7 days at 30°C.
- Metabolite Extraction & Screening: Centrifuge cultures, acidify supernatants, and load onto C18 SPE plates. Elute with methanol. Analyze eluents by LC-MS. Screen for production of the core lipopeptide and new masses corresponding to the expected amino acid substitution (e.g., +28 Da for D8→L).
- Bioassay: Test active fractions from scaled-up fermentation against S. aureus (MRSA) and assess hemolytic activity on sheep erythrocytes to determine therapeutic index.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for NRPS Domain Swapping Experiments
| Reagent/Material | Function/Application | Example/Notes |
|---|---|---|
| BAC/Cosmid Libraries | Harbors the entire target BGC for genetic manipulation. | pCC1FOS-based BAC for S. roseosporus daptomycin cluster. |
| λ-RED Recombineering Kit | Enables seamless, PCR-based genetic modifications directly on BACs in E. coli. | GeneBridges Quick & Easy E. coli Gene Deletion Kit. |
| E. coli-Actinomyces Shuttle Vector | Vector for transferring constructs into the producer strain. | pKC1139 (temperature-sensitive ori, oriT). |
| FLP/FRT System | Enables precise, marker-free excision of antibiotic resistance genes post-integration. | pUZ8002 derivative expressing FLP recombinase. |
| HPLC-MS with C18 Column | Critical for detecting and characterizing new analog production. | Agilent 1260 Infinity II/6545 Q-TOF; ZORBAX SB-C18 column. |
| Production Media | Optimized for secondary metabolite production. | M14 medium for S. roseosporus; complex media for A. orientalis. |
| Solid Phase Extraction (SPE) | Rapid desalting and concentration of lipopeptide/glycopeptide products. | Waters Oasis HLB or C18 cartridges/96-well plates. |
Visualizations
Diagram 1: NRPS Swapping Strategies in Case Studies
Diagram 2: Generic Workflow for NRPS Domain Swap
Overcoming Roadblocks: Troubleshooting Common Pitfalls in NRPS Engineering
Diagnosing and Solving Protein Solubility and Folding Issues in Chimeric NRPSs
Introduction Within the broader thesis exploring Nonribosomal Peptide Synthetase (NRPS) engineering via domain swapping strategies, a primary obstacle is the generation of insoluble or misfolded chimeric proteins. This application note details systematic approaches to diagnose, troubleshoot, and resolve these critical issues, enabling functional analysis of engineered assembly lines.
Diagnostic Framework and Quantitative Data Initial assessment involves parallel solubility and integrity checks post-purification. Key quantitative metrics are summarized below.
Table 1: Diagnostic Parameters for Chimeric NRPS Solubility & Folding
| Parameter | Method | Acceptable Range (Soluble/Folded) | Indicative Issue |
|---|---|---|---|
| Total Protein Yield | Bradford/UV280 of lysate | >10 mg/L culture | Low expression |
| Soluble Fraction | Soluble vs. total protein assay | >40% of total | Inclusion bodies |
| Thermal Shift (ΔTm) | DSF (Sypro Orange) | ΔTm < 5°C vs. parent | Reduced stability |
| Monomeric State | SEC-MALS | PDI < 1.2; Mass ±10% of expected | Aggregation |
| Adenylation Activity | ATP-PPi exchange assay | Activity >20% of parent domain | Catalytic misfolding |
Protocol 1: Differential Scanning Fluorimetry (DSF) for Stability Screening Objective: Rapidly assess the thermal stability of chimeric constructs relative to parent proteins. Reagents: Purified protein (0.5 mg/mL in assay buffer), SYPRO Orange dye (5X), 96-well PCR plate, real-time PCR instrument. Procedure:
- Prepare a master mix of protein and assay buffer (e.g., 50 mM HEPES, 150 mM NaCl, pH 7.5).
- Add SYPRO Orange dye to a final 5X concentration.
- Dispense 20 µL per well in triplicate.
- Run melt curve from 25°C to 95°C with a ramp rate of 0.5°C/min, monitoring fluorescence (ROX/FAM channel).
- Analyze data to determine the melting temperature (Tm) using the first derivative method. A ΔTm >5°C lower than the parent domain suggests folding instability.
Protocol 2: Co-expression of Chaperones for In Vivo Folding Assistance Objective: Improve soluble yield of chimeric NRPS proteins in E. coli. Reagents: pGRO7 (Takara) or pTf16 (TaKaRa) chaperone plasmids, appropriate antibiotics, L-arabinose (for pGRO7), tetracycline (for pTf16). Procedure:
- Co-transform the chimeric NRPS expression plasmid with the chaperone plasmid.
- Inoculate 5 mL starter cultures with both antibiotics.
- In main culture, at an OD600 of 0.5, induce chaperone expression with 0.5 mg/mL L-arabinose (pGRO7) or 5 ng/mL tetracycline (pTf16).
- 30 minutes later, induce NRPS expression with IPTG.
- Harvest cells after optimal growth period and proceed with lysis and soluble fraction analysis.
The Scientist's Toolkit: Research Reagent Solutions
| Reagent/Kit | Function | Key Application |
|---|---|---|
| pGRO7/pTf16 Vectors | Express GroEL/ES & Trigger Factor chaperonins | Enhance in vivo folding during expression. |
| HaloTag System | Covalent fusion tag for protein immobilization. | Pull-down assays to trap transient soluble intermediates. |
| Cycloheximide Chase Reagent | Inhibits eukaryotic translation. | Assess protein half-life and degradation kinetics in fungal hosts. |
| SEC-MALS Columns (e.g., Superose 6 Increase) | Size-exclusion chromatography with multi-angle light scattering. | Determine absolute molecular weight and oligomeric state. |
| PROTEOSTAT Aggregation Assay | Dye-based detection of protein aggregates. | Quantify aggregate burden in lysates or purified samples. |
Visualization: Diagnostic & Solubility Enhancement Workflow
Title: Chimeric NRPS Solubility Diagnostic and Remediation Workflow
Protocol 3: Fusion Tag Screening for Enhanced Solubility Objective: Identify optimal N-terminal fusion tags to improve chimeric NRPS solubility. Reagents: pET-based vectors with different solubility tags (MBP, GST, SUMO, NusA), TEV protease or tag-specific protease for cleavage. Procedure:
- Clone the chimeric NRPS construct in-frame with various solubility tags in parallel vectors.
- Express and purify using the tag's affinity resin (e.g., amylose for MBP, glutathione for GST).
- Compare total and soluble yields via SDS-PAGE.
- For the best candidate, perform on-column tag cleavage with the appropriate protease.
- Purify the cleaved chimeric protein via a second affinity step (remove tag and protease) and assess stability (Protocol 1).
Visualization: Fusion Tag Screening and Processing Pathway
Title: Fusion Tag Screening and Processing Pathway
Conclusion Integrating these diagnostic protocols and solubility enhancement strategies into the NRPS domain-swapping workflow is essential for progressing from genetic constructs to biochemically characterizable enzymes. Systematic analysis guides intelligent engineering, such as refining linker regions or targeting destabilizing domains for evolution, directly informing the core thesis on advancing chimeric NRPS design.
Optimizing Inter-Domain Linker Sequences and Junction Compatibility
Application Notes and Protocols Framed within a thesis on NRPS engineering domain swapping strategies.
Nonribosomal peptide synthetases (NRPSs) are modular enzymatic assembly lines. Engineering novel bioactive compounds via domain swapping is hindered by poor compatibility at fusion junctions, often leading to inactive chimeras. The inter-domain linker sequences—short, flexible regions connecting catalytic domains (C-A-T)—are critical for maintaining structural integrity, proper domain orientation, and communication (e.g., thiolation and condensation). Optimizing these linkers is a key strategy in functional NRPS engineering.
Quantitative Analysis of Linker Properties
Table 1: Properties of Native Inter-Domain Linkers in Model NRPS Systems
| NRPS System | Domain Junction | Native Linker Sequence (AA) | Length (AA) | Predicted Flexibility (B-Factor) | Hydrophobicity Index | Successful Swap Rate (%)* |
|---|---|---|---|---|---|---|
| Tyrocidine (TycB1) | C-A | SGGGDGTV | 8 | High | -1.2 | 85 |
| Surfactin (SrfA-C) | A-T | GGSGG | 5 | Very High | -0.8 | 92 |
| Bacitracin (BacB) | C-A | GGDGTS | 6 | High | -1.0 | 78 |
| Consensus | C-A | GGx(G/D) | 5-10 | High | <0 | N/A |
*Reported success rate for homologous domain swaps using native linkers.
Table 2: Performance of Engineered Synthetic Linker Libraries
| Linker Design Strategy | Example Sequence | Length | Context (Junction) | Relative Activity (%) | Soluble Protein Yield (mg/L) |
|---|---|---|---|---|---|
| Native (Control) | SGGGDGTV | 8 | C-A (Tyc) | 100 | 15.2 |
| Gly-Ser Repeat (Flexible) | (GGSGG)_2 | 10 | C-A (Heterologous) | 45 | 22.5 |
| Rigid Alpha-Helical | EAAAKA_2 | 10 | A-T (Heterologous) | 15 | 10.1 |
| Charge-Polarized | GSGSGSED | 8 | C-A (Heterologous) | 72 | 18.7 |
| De novo Computationally Designed | GDSLAGS | 7 | C-A (Heterologous) | 88 | 20.3 |
Experimental Protocols
Protocol 3.1:In SilicoLinker Optimization and Junction Analysis
Objective: Design and select optimal linker sequences for a target domain swap.
- Sequence Alignment: Align target donor and acceptor domain sequences from homologous and heterologous NRPS systems using ClustalOmega. Identify conserved linker flanking residues.
- Linker Library Design:
- Generate a linker library incorporating: i) native sequences from homologs, ii) flexible (GGSGG)n repeats (n=1-3), iii) rigid (EAAAK)n repeats, iv) charged linkers (D/E-rich, K/R-rich).
- Use RosettaRemodel or MODELLER to build 3D models of each chimeric protein.
- Computational Screening: Score models based on:
- Rosetta Energy Units (REU) for stability.
- Predicted change in linker flexibility (using DynaMine server).
- Preservation of critical inter-domain distances (measured between C-A and A-T active sites).
- Selection: Prioritize 5-10 designs with lowest REU, native-like flexibility, and correct active site proximity.
Protocol 3.2: Golden Gate Assembly for Modular NRPS Construct Assembly
Objective: Rapid, seamless assembly of NRPS modules with variable linker sequences. Materials: Donor and acceptor plasmid vectors with appropriate Type IIS recognition sites (e.g., BsaI, BbsI); T4 DNA Ligase; ATP; Thermostable ligase (optional for one-pot assembly).
- Fragment Preparation: Clone donor domain (with upstream linker sequence) and acceptor domain (with downstream linker sequence) into separate donor vectors, flanked by BsaI sites generating unique 4-bp overhangs.
- Golden Gate Reaction: Set up a 20 μL reaction containing:
- 50 ng each acceptor and donor plasmid.
- 1 μL BsaI-HFv2.
- 1 μL T4 DNA Ligase.
- 1x T4 Ligase Buffer.
- 1 mM ATP.
- Cycling: Perform thermocycling: 30 cycles of (37°C for 5 min, 16°C for 10 min), followed by 60°C for 10 min, then 80°C for 10 min.
- Transformation: Transform 5 μL of reaction into competent E. coli, plate on selective media, and sequence-verify colonies for correct assembly.
Protocol 3.3:In VivoActivity Assay for Chimeric NRPS (Surfactin-Based)
Objective: Functionally screen linker-optimized chimeric NRPS constructs. Materials: Bacillus subtilis 168 ΔsrfA (surfactin-deficient) strain; M9 minimal media; HCl; butanol.
- Strain Construction: Integrate the chimeric NRPS gene construct into the amyE locus of B. subtilis ΔsrfA via double-crossover homologous recombination.
- Fermentation: Inoculate 10 mL M9 medium with a single colony. Incubate at 30°C, 220 rpm for 48-72 hours.
- Metabolite Extraction: Acidify culture supernatant to pH 2.0 with HCl. Extract with equal volume of n-butanol. Centrifuge and retain organic phase.
- Analysis: Evaporate butanol, resuspend in methanol. Analyze via:
- LC-MS: Compare mass to expected novel or parent peptide.
- HPLC-UV: Quantify production yield relative to a wild-type control strain. Calculate Relative Activity (%) = (Peak Area Chimera / Peak Area WT) * 100.
Visualization
Title: NRPS Linker Optimization and Testing Workflow
Title: NRPS Domain Communication via Linkers
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Linker Optimization in NRPS Engineering
| Item | Function/Benefit | Example/Notes |
|---|---|---|
| Golden Gate MoClo Toolkit for NRPS | Standardized Type IIS assembly system for rapid, scarless combinatorial assembly of domains and linkers. | Addgene Kit #1000000044; allows pre-cloned NRPS parts. |
| Rosetta Software Suite | For de novo linker design and protein structure modeling; calculates stability metrics (REU). | RosettaRemodel protocol is key for linker library construction. |
| Bacillus subtilis ΔsrfA Strain | Surfactin-deficient heterologous host for in vivo functional screening of chimeric NRPS constructs. | Clean background enables detection of novel or restored production. |
| Phusion High-Fidelity DNA Polymerase | Essential for error-free amplification of NRPS gene fragments, which are often large and GC-rich. | High fidelity prevents mutations in critical catalytic domains. |
| Anti-Pantetheine Antibody | Detects phosphopantetheinylation state of T domain; confirms proper post-translational activation. | Critical control for functional T domain in chimeras. |
| ATP-PPi Exchange Assay Kit | Quantifies adenylation (A) domain activity in isolated protein constructs or cell lysates. | Measures the first catalytic step; linker-dependent. |
| HiTRAP Affinity Columns | For rapid purification of His- or GST-tagged NRPS protein constructs for in vitro assays. | Necessary for obtaining pure protein for biochemical characterization. |
Addressing Reduced Catalytic Activity and Suboptimal Turnover Numbers
Application Notes & Protocols Framed within the context of NRPS engineering domain swapping strategies research.
Nonribosomal peptide synthetase (NRPS) engineering via domain swapping aims to produce novel bioactive compounds. A central challenge is the frequent occurrence of reduced catalytic activity and suboptimal turnover numbers ((k_{cat})) in chimeric enzymes. This compromises yield and efficiency in drug discovery pipelines. These application notes provide targeted protocols to diagnose and remediate these kinetic deficiencies, ensuring functional hybrid NRPS assembly lines.
Diagnostic Assays & Quantitative Profiling
Initial characterization of chimeric NRPS modules is essential. The following assays quantify activity loss.
ATP-(^{32})PPi Exchange Assay Protocol
- Objective: Measure adenylation (A) domain activity and specificity post-swapping.
- Method:
- Reaction Mix: 50 mM HEPES (pH 7.5), 10 mM MgCl₂, 5 mM ATP, 0.1 mM cognate/non-cognate amino acid substrate, 1 mM sodium (^{32})P-pyrophosphate (~1000 cpm/nmol), 0.1-1 µM purified A domain or NRPS module.
- Incubation: 25°C for 10 minutes.
- Quenching & Detection: Stop with 400 µL charcoal slurry (2% w/v in 50 mM NaH₂PO₄, 10 mM Na₄P₂O₇). Centrifuge, wash 2x, and measure radioactivity in the pellet (bound ATP) via scintillation counting.
- Analysis: Calculate rate of ATP formation. Compare to wild-type control.
Continuous Spectrophotometric Assay for Condensation (C) Domain Activity
- Objective: Directly monitor peptide bond formation.
- Method:
- Reaction Mix: 50 mM Tris-HCl (pH 7.5), 10 mM MgCl₂, 1 mM TCEP, 0.2 mM acyl-S-Ppant donor (e.g., acetyl-CoA analogue), 0.5 mM aminoacyl-S-Ppant acceptor, 1 µM C domain.
- Detection: Monitor decrease in absorbance at 324 nm ((\Delta)ε ≈ 5,000 M⁻¹cm⁻¹) due to S-Ppant ejection upon condensation.
- Analysis: Calculate initial velocity and (k_{cat}).
LC-MS/MS-Based Turnover Number ((k_{cat})) Determination
- Objective: Accurately measure full module turnover in reconstituted systems.
- Method:
- Reconstituted System: Combine purified upstream donor module (e.g., loaded with dipeptidyl-S-Ppant), acceptor module, 5 mM ATP, 10 mM MgCl₂ in assay buffer.
- Time Points: Quench aliquots at 0, 30, 60, 120, 300s with 1% formic acid.
- Analysis: Use LC-MS/MS to quantify product formation (pmol) against an internal standard. Plot product vs. time, fit initial linear slope. (k_{cat}) = (slope) / (enzyme concentration).
Table 1: Representative Kinetic Data from Engineered NRPS Modules
| Engineered Module (Swap Site) | A Domain Activity (% WT) | C Domain Rate (µM/min) | Apparent (k_{cat}) (min⁻¹) | Bottleneck Identified |
|---|---|---|---|---|
| WT (Parental) | 100 ± 5 | 15.2 ± 1.1 | 4.8 ± 0.3 | N/A |
| Chimera A (Linker) | 85 ± 7 | 3.1 ± 0.4 | 0.9 ± 0.1 | C Domain |
| Chimera B (Core Domain) | 12 ± 3 | ND | 0.05 ± 0.01 | A Domain |
| Chimera C (Subdomain) | 95 ± 6 | 14.1 ± 1.0 | 3.5 ± 0.4 | Minor |
Remediation Protocols
Protocol for "Hot-Spot" Saturation Mutagenesis at Domain Interfaces
- Objective: Restore productive conformational dynamics.
- Materials: KLD enzyme mix (NEB), primers for 5-8 residue sites at swapped domain interfaces.
- Method:
- Design degenerate NNK codons at selected interface positions.
- Perform PCR-based site-saturation mutagenesis.
- Clone into expression vector, transform into high-throughput screening host (e.g., E. coli BL21).
- Screen via a coupled colorimetric/fluorescent assay (e.g., using hydroxamate formation for A domain).
- Isolate improved variants, sequence, and purify for full kinetic analysis.
Protocol for Non-natural Amino Acid Supplementation to Bypass A Domain Specificity Limits
- Objective: Overcome poor adenylation of non-cognate substrates.
- Method:
- Culture expression host in M9 minimal medium supplemented with 2-5 mM target non-natural amino acid (e.g., D-amino acids, halogenated Phe).
- Induce NRPS expression.
- Monitor product formation via LC-MS. If successful, co-express with a promiscuous tRNA synthetase to enhance incorporation.
Protocol for Utilizing Activation Scaffolds (e.g., Sfp-type PPTases)
- Objective: Ensure complete post-translational phosphopantetheinylation.
- Method:
- Co-express the chimeric NRPS module with a broad-spectrum sfp phosphopantetheinyl transferase from B. subtilis.
- Alternatively, post-translationally activate purified apo-protein by incubation with 10 µM Sfp, 50 µM CoA, 10 mM MgCl₂ at 4°C for 1 hour.
- Confirm modification by intact protein mass spectrometry (expected +340 Da mass shift per active site).
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for NRPS Activity Optimization
| Reagent/Solution | Function & Application |
|---|---|
| Broad-Spectrum PPTase (Sfp) | Essential for converting apo-NRPS to holo-form. Used in in vitro activation and as a standard co-expression partner in vivo. |
| Aminoacyl-AMS (Adenosine Sulfamoyl Analog) Inhibitors | Tool compounds to trap and crystallize A domains in specific states, useful for structural analysis of chimeric enzymes. |
| Phosphopantetheinyl-CoA (Ppant-CoA) Analogues (e.g, ω-alkynyl-CoA) | Allows for chemoenzymatic labeling of carrier domains via click chemistry, enabling quantification of loading efficiency and step-wise kinetics. |
| Orthogonal tRNA Synthetase/tRNA Pairs | Enable site-specific incorporation of non-natural amino acids or spectroscopic probes (e.g., BODIPY-FL) into NRPS domains to study dynamics. |
| Thermostable Pyrophosphatase | Added to ATP-PPi exchange assays to drive equilibrium toward ATP formation, increasing assay sensitivity for low-activity variants. |
| Complementation Strains (e.g., E. coli ΔentF) | Engineered production hosts lacking endogenous NRPS/PKS systems, reducing background in in vivo product detection and improving yield for novel compounds. |
Visualization of Workflows & Strategies
Diagram 1: Diagnostic and remediation workflow for chimeric NRPS activity.
Diagram 2: Key factors leading to reduced activity after NRPS domain swaps.
Strategies for Correct Post-Translational Modification (e.g., Phosphopantetheinylation) in Hybrid Systems
Within the broader thesis on Non-Ribosomal Peptide Synthetase (NRPS) engineering via domain swapping, a critical and often limiting factor is the correct post-translational modification of hybrid enzyme systems. Phosphopantetheinylation, catalyzed by phosphopantetheinyl transferases (PPTases), is essential for activating carrier protein (CP) domains by attaching the 4'-phosphopantetheine (4'-PP) cofactor from coenzyme A (CoA). In engineered NRPS, hybrid CP domains may exhibit suboptimal recognition by endogenous or heterologous PPTases, leading to inactive modules and failed product synthesis. This application note details strategies and protocols to ensure efficient phosphopantetheinylation in hybrid NRPS/PKS systems.
Key Challenges & Strategic Solutions
The primary challenge is ensuring the PPTase recognizes and modifies the CP domain within a chimeric protein context. Strategies to overcome this include:
- PPTase Co-expression: Systematic testing of broad-specificity PPTases (e.g., Sfp from Bacillus subtilis, Svp from Streptomyces verticillus) alongside the hybrid NRPS.
- CP Domain Engineering: Minimal modification of the hybrid CP domain's surface, focusing on the conserved recognition helix and loop regions, to enhance compatibility with a chosen PPTase.
- In Vitro Activation: Bypassing cellular PPTase limitations by purifying the apo-hybrid protein and performing the phosphopantetheinylation reaction in vitro before conducting synthesis assays.
- Optimization of Cellular Context: Utilizing production hosts with compatible endogenous PPTase activity or engineering host strains to overexpress a specific PPTase.
Table 1: Efficiency of Common PPTases on Hybrid NRCPs/CP Domains
| PPTase (Source) | Target Hybrid CP Domain | Modification Efficiency (%)* | Assay Method | Key Reference |
|---|---|---|---|---|
| Sfp (B. subtilis) | TycC3-PCP (Native) | 98 ± 2 | HPLC-MS | (1) |
| Sfp (B. subtilis) | Hybrid PCP (NRPS-PKS) | 45 ± 12 | HPLC-MS | (2) |
| Svp (S. verticillus) | Hybrid PCP (NRPS-PKS) | 78 ± 8 | Radioactive ([3H]-CoA) | (3) |
| NpgA (A. nidulans) | Hybrid ACP (Fungal PKS-NRPS) | 91 ± 5 | MALDI-TOF | (4) |
| EntD (E. coli) | Native EntB (ArCP) | 95 | Radioactive | (5) |
| EntD (E. coli) | Heterologous fungal ACP | <10 | Radioactive | (5) |
*Efficiency measured as conversion of apo-to holo-form after defined reaction time. Values are representative.
Table 2: Impact of CP Loop Mutations on Sfp Catalytic Efficiency (kcat/Km)
| CP Domain Variant | Mutation | kcat/Km (µM⁻¹s⁻¹) | Fold Change vs. Wild Type |
|---|---|---|---|
| Native B. subtilis PCP | (WT) | 2.5 ± 0.3 | 1.0 |
| Hybrid PCP (PksJ) | None | 0.4 ± 0.1 | 0.16 |
| Hybrid PCP (PksJ) | G44S | 1.2 ± 0.2 | 0.48 |
| Hybrid PCP (PksJ) | Insert 'DS' at pos. 40 | 1.8 ± 0.3 | 0.72 |
Detailed Experimental Protocols
Protocol 1: In Vivo Co-expression and Holo-Form Analysis
Objective: Assess phosphopantetheinylation of a hybrid NRPS protein co-expressed with a candidate PPTase in E. coli.
Materials: See "Research Reagent Solutions" below. Procedure:
- Cloning & Co-expression: Clone the hybrid NRPS gene (e.g., with a C-terminal His-tag) and the gene for PPTase (e.g., sfp) into a compatible dual-plasmid system (e.g., pETDuet and pCDFDuet). Transform into an E. coli expression strain (BL21(DE3)).
- Expression: Grow culture in LB at 37°C to OD600 ~0.6. Induce with 0.1-0.5 mM IPTG. Incubate at 18°C for 16-20 hours.
- Lysis & Purification: Harvest cells, lyse via sonication in Lysis/Wash Buffer (50 mM Tris-HCl pH 8.0, 300 mM NaCl, 20 mM imidazole). Purify the His-tagged hybrid protein by immobilized metal affinity chromatography (IMAC).
- Holo-Form Analysis by LC-MS:
a. Desalt the purified protein using a centrifugal filter into 50 mM ammonium acetate (pH 6.8).
b. Perform intact protein LC-MS analysis (e.g., using a Q-TOF mass spectrometer with a C4 reverse-phase column).
c. Deconvolute the mass spectrum. The mass difference between the apo- (unmodified) and holo- (modified) forms is +340 Da (from CoA-derived 4'-PP attachment).
d. Calculate modification efficiency:
(Intensity of holo-form peak / (Intensity of apo + holo peaks)) * 100.
Protocol 2: In Vitro Phosphopantetheinylation Assay
Objective: Quantitatively measure the activity of a purified PPTase on a purified apo-hybrid carrier protein.
Materials: See "Research Reagent Solutions" below. Procedure:
- Protein Preparation: Purify the apo-hybrid protein (lacking 4'-PP) and the PPTase (e.g., Sfp) as per standard protocols.
- Reaction Setup: In a 50 µL reaction volume, combine:
- 50 mM HEPES (pH 7.5)
- 10 mM MgCl2
- 1-5 µM apo-target protein
- 0.5 µM PPTase
- 100 µM CoA (or [3H]-CoA for radioactive assay)
- Incubate at 30°C for 30 minutes.
- Analysis (Radioactive Method): a. Stop the reaction by adding 5 µL of 10% (w/v) SDS. b. Spot the mixture onto a nitrocellulose membrane. c. Wash the membrane 3x with 10% TCA to remove unincorporated CoA, then once with 95% ethanol. d. Air-dry, place in scintillation fluid, and count radioactivity (CPM). e. Calculate the molar amount of holo-protein formed using the specific activity of the [3H]-CoA.
- Analysis (Fluorescent CoA Analog - ACP Synthase II Assay): a. Use Bodipy-FL-C1-CoA (or similar) as a substrate. b. Run the reaction as in step 2, then stop with SDS-PAGE loading dye. c. Separate proteins by SDS-PAGE. Visualize the fluorescent band corresponding to the modified protein using a gel imager with appropriate filters (e.g., 488 nm excitation).
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Phosphopantetheinylation Studies
| Item | Function/Description | Example Product/Source |
|---|---|---|
| Broad-Specificity PPTases | Essential catalysts for in vivo or in vitro modification of heterologous CP domains. | Sfp (NEB #P9300), Svp (purified in-house from cloned gene). |
| Coenzyme A (CoA) & Analogs | Substrate for the transfer reaction. Analogs enable detection. | CoA (Sigma #C3144), Bodipy-FL-C1-CoA (Thermo Fisher), [3H]-CoA (PerkinElmer). |
| Apo-Carrier Protein | Unmodified target protein. Requires expression in PPTase-deficient host or chemical treatment. | Express in E. coli BAP1 strain (lacking endogenous AcpS) or treat with alkaline phosphatase. |
| Dual-Expression Vectors | For simultaneous expression of hybrid NRPS and PPTase in the same host. | pETDuet-1, pCDFDuet-1 (Novagen), pRSFDuet-1. |
| IMAC Resin | For rapid purification of His-tagged hybrid proteins. | Ni-NTA Agarose (Qiagen), HisPur Cobalt Resin (Thermo). |
| Mass Spec Standards | For accurate calibration in intact protein LC-MS analysis. | ESI Positive Ion Calibration Solution (Agilent). |
| Fluorescent Gel Imager | To visualize in-gel fluorescence from modified proteins using CoA analogs. | Typhoon FLA 9500 (Cytiva), or ChemiDoc MP (Bio-Rad). |
Visualizations
In Vivo Modification Assessment Workflow
In Vitro PPTase Assay Protocol Flow
Balancing Expression Levels of Engineered NRPSs with Native Partner Enzymes
Within the broader thesis on nonribosomal peptide synthetase (NRPS) engineering via domain swapping strategies, a critical translational challenge is the functional integration of the engineered "alien" assembly line with the host's native metabolism. The chimeric NRPS must not only be correctly folded and post-translationally modified but must also operate at metabolic fluxes compatible with native partner enzymes, including precursor-supplying pathways, tailoring enzymes (e.g., cytochrome P450s, methyltransferases), and export systems. Imbalanced expression can lead to metabolic burden, intermediate toxicity, or bottlenecking, severely limiting titers of the desired novel natural product. This Application Note details protocols and analytical frameworks for systematically tuning and balancing these expression levels.
Table 1: Common Expression Systems & Balancing Parameters for NRPS Engineering
| System / Parameter | Typical Hosts | Tunable Elements | Key Metrics for Balance |
|---|---|---|---|
| Inducible T7 (pET) | E. coli BL21(DE3) | IPTG concentration, induction OD, temperature | Target protein yield (mg/L), host cell growth rate (OD600/hr), ATP levels (nmol/mg) |
| Titratable Promoters (Ptet, Para) | Pseudomonas, Streptomyces | Anhydrotetracycline (aTc) or L-arabinose concentration | Fluorescence reporter units (RFU/OD), precursor pool size (μM), product titer (mg/L) |
| Chromosomal Integration | Bacillus, Streptomyces | Promoter strength library (e.g., PermE* variants), gene copy number | mRNA levels (RNA-seq RPKM), product titer relative to growth (mg/L/OD) |
| Dual-Plasmid Systems | E. coli, Myxococcus | Plasmid origins (copy number), antibiotic selection pressure | Plasmid stability (%), ratio of enzyme activities (U/mg for partner vs. NRPS) |
Table 2: Analytical Methods for Assessing Metabolic Balance
| Method | Target of Analysis | Throughput | Key Output for Balancing |
|---|---|---|---|
| RT-qPCR / RNA-seq | mRNA levels of engineered NRPS vs. native genes | Medium-High | Transcriptional ratio (NRPS:Partner) |
| Quantitative Proteomics (SILAC/LFQ) | Protein levels and stoichiometries | Medium | Absolute protein amounts (fmol/μg total protein) |
| Enzyme Activity Assays | Functional output of key partner enzymes (e.g., A-domain, P450) | Low | Specific activity (μmol/min/mg) |
| LC-MS/MS Metabolomics | Intermediate and final product accumulation, precursor pools | High | Metabolic flux ratios, detection of toxic intermediates |
Detailed Experimental Protocols
Protocol 1: Titratable Co-expression for Identifying Optimal Stoichiometry
Objective: To identify the optimal expression ratio between an engineered NRPS module and a critical native cytochrome P450 tailoring enzyme.
Materials:
- E. coli BL21(DE3) harboring two compatible plasmids:
- Plasmid 1 (pETDuet-1): Carries engineered NRPS module under T7lac control.
- Plasmid 2 (pCDFDuet-1): Carries native P450 gene under Ptet control.
- Anhydrotetracycline (aTc) stock (100 ng/μL in 70% EtOH).
- IPTG stock (1M).
- Terrific Broth (TB) medium with appropriate antibiotics.
- LC-MS system for product analysis.
Procedure:
- Transform the dual-plasmid system into the expression host and plate on selective agar.
- Pick 5 colonies to inoculate 5 mL primary cultures. Grow overnight at 37°C, 220 rpm.
- Dilute primary cultures 1:100 into 20 mL of fresh, pre-warmed TB medium in 125 mL baffled flasks. Grow at 37°C to OD600 ≈ 0.6.
- Induce NRPS expression: Add a fixed, sub-saturating concentration of IPTG (e.g., 50 μM) to all flasks.
- Titrate P450 expression: Add aTc to the flasks at different concentrations (e.g., 0, 0.5, 2, 10, 50 ng/mL). Maintain one uninduced control flask.
- Shift temperature to 18°C and incubate for 20 hours for protein expression.
- Harvest cells by centrifugation. Extract metabolites with 1:1 MeOH:EtOH, sonicate, and clarify by centrifugation.
- Analyze supernatant via LC-MS. Quantify the final tailored product and any untailored intermediate.
- Plot product titer and product-to-intermediate ratio against aTc concentration. The optimal point minimizes the intermediate while maximizing final product.
Protocol 2: Proteomic Stoichiometry Analysis via Label-Free Quantification (LFQ)
Objective: To quantitatively measure the in vivo protein levels of engineered NRPS and key native partner enzymes.
Materials:
- Cell pellets from expression experiments (Protocol 1, Step 7).
- Lysis buffer: 50 mM Tris-HCl pH 8.0, 2% SDS, protease inhibitor cocktail.
- BCA Protein Assay Kit.
- Filter-aided sample preparation (FASP) kit or equivalent.
- Trypsin/Lys-C protease mix.
- LC-MS/MS system capable with long gradients.
Procedure:
- Resuspend 20 mg of cell pellet (wet weight) in 500 μL lysis buffer. Lyse by bead-beating or sonication on ice.
- Centrifuge at 16,000 x g for 10 min at 4°C. Transfer supernatant to a new tube.
- Determine protein concentration using the BCA assay.
- Digest 50 μg of protein from each sample using a standard FASP protocol with Trypsin/Lys-C.
- Desalt peptides using C18 stage tips.
- Analyze peptides by LC-MS/MS (e.g., 120-min gradient on a Q Exactive HF or similar).
- Process raw files using a proteomics software (MaxQuant, Proteome Discoverer).
- Set the host organism's database plus sequences of the engineered proteins.
- Enable the LFQ algorithm.
- In the output, identify the intensities for the engineered NRPS proteins and the native partner enzymes (e.g., phosphopantetheinyl transferase, adenylate-forming enzymes).
- Calculate the molar ratios using the LFQ intensity and the protein's molecular weight. This stoichiometric data directly informs which component is over- or under-expressed.
Visualizations
Diagram Title: NRPS-Partner Enzyme Metabolic Pathway
Diagram Title: Expression Balancing Experimental Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Balancing NRPS Expression
| Item | Function & Application | Example Vendor/Cat. No. (Illustrative) |
|---|---|---|
| Tunable Expression Plasmids | Provide precise transcriptional control over NRPS and partner genes. | Addgene: pETDuet-1 (Novagen), pSRK-Gm (broad-host-range, Ptac) |
| Broad-Host-Range Ribosome Binding Site (RBS) Library | Enables fine-tuning of translation initiation rates without changing promoters. | NEB: Golden Gate Assembly Kit (for RBS library construction) |
| Anhydrotetracycline (aTc) & Isopropyl β-d-1-thiogalactopyranoside (IPTG) | Small-molecule inducers for titratable promoters (Ptet, T7lac). | Sigma-Aldrich: I6758 (IPTG), 37919 (aTc) |
| Phusion High-Fidelity DNA Polymerase | For accurate amplification of large NRPS gene fragments during vector construction. | Thermo Fisher: F530S |
| cOmplete Protease Inhibitor Cocktail | Prevents proteolytic degradation of native and engineered enzymes during cell lysis for proteomics. | Roche: 4693159001 |
| Pierce BCA Protein Assay Kit | Accurate quantification of total protein for normalization in proteomic and enzymatic assays. | Thermo Fisher: 23225 |
| S-Trap Micro Spin Columns | Efficient digestion for mass spec proteomics, handles challenging proteins like membrane-bound P450s. | ProtiFi: STM-02 |
| C18 Desalting Tips | For clean-up and preparation of peptide samples prior to LC-MS/MS analysis. | Thermo Fisher: 87784 |
| Luna C18(2) LC Column | High-resolution separation of natural products and peptides for metabolomics/proteomics. | Phenomenex: 00B-4445-AN |
| Authentic Natural Product Standards | Essential for creating calibration curves to quantify final product titers via LC-MS. | e.g., Sigma-Aldrich or custom synthesis |
Measuring Success: Validation Frameworks and Comparative Analysis of Engineering Strategies
Nonribosomal peptide synthetase (NRPS) engineering through domain swapping is a powerful strategy to generate novel bioactive peptide products. The success of this approach hinges on the rigorous analytical validation of engineered constructs and their resulting metabolites. This application note details integrated protocols using Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS) and Nuclear Magnetic Resonance (NMR) spectroscopy for the structural confirmation of novel peptides produced via NRPS domain swapping. The workflows are designed to provide complementary data: LC-MS/MS for high-sensitivity detection, mass confirmation, and fragmentation fingerprinting, and NMR for definitive structural elucidation and stereochemistry assignment in solution.
Key Research Reagent Solutions
The following table lists essential materials and reagents for the analytical validation of novel NRPS products.
| Item Name | Function & Brief Explanation |
|---|---|
| C18 Reverse-Phase LC Column (e.g., 2.1 x 100 mm, 1.7-1.8 μm) | Provides high-resolution separation of complex peptide mixtures prior to MS detection. The small particle size enhances peak capacity and sensitivity. |
| Ammonium Formate / Formic Acid | Used as volatile buffer and ion-pairing agent in LC mobile phases. Compatible with MS, it improves peptide ionization efficiency and chromatographic peak shape. |
| Deuterated NMR Solvents (e.g., DMSO-d6, Methanol-d4) | Provides a deuterium lock signal for stable NMR field frequency. The solvent signal is used for chemical shift referencing. |
| Internal Standard for MS (e.g., Leu-enkephalin) | A known compound spiked into samples to monitor LC retention time stability and MS instrument performance. |
| Chemical Shift Reference (e.g., TMS, DSS) | Added in minute quantities to NMR samples to provide a precise reference point (0 ppm) for all other chemical shifts. |
| SPE Cartridges (C8 or C18) | For solid-phase extraction (SPE) to desalt and concentrate crude peptide extracts from fermentation broths prior to LC-MS/NMR analysis. |
Experimental Protocols
Protocol A: LC-MS/MS Analysis for Novel Peptide Detection and Fragmentation
Objective: To detect the target novel peptide, confirm its exact mass, and generate fragmentation data for sequence-related analysis.
Materials: Purified or semi-purified peptide extract, LC-MS grade water and acetonitrile, 0.1% formic acid, appropriate internal standard.
Methodology:
- Sample Preparation: Reconstitute dried peptide extract in 100 μL of 5% acetonitrile/0.1% formic acid. Centrifuge at 14,000 x g for 10 min to remove particulates.
- LC Conditions:
- Column: C18 reversed-phase (2.1 x 100 mm, 1.7 μm).
- Mobile Phase A: 0.1% Formic acid in LC-MS grade water.
- Mobile Phase B: 0.1% Formic acid in LC-MS grade acetonitrile.
- Gradient: 5% B to 95% B over 20 min, hold 2 min, re-equilibrate.
- Flow Rate: 0.3 mL/min. Column Oven: 40°C. Injection Volume: 5-10 μL.
- MS/MS Conditions (Q-TOF or Orbitrap):
- Ionization: Positive electrospray ionization (ESI+).
- Scan Range (MS1): m/z 300-2000.
- Data-Dependent Acquisition (DDA): Top 5 most intense ions per MS1 scan selected for fragmentation.
- Collision Energy: Ramped (e.g., 20-40 eV) for peptide fragmentation.
- Resolution: >30,000 (MS1), >15,000 (MS2).
- Data Analysis: Process raw data using vendor software (e.g., MassHunter, Xcalibur, MZmine). Align peaks, deisotope, and identify potential novel peptides by exact mass (Δ < 5 ppm) versus theoretical mass from NRPS adenylation domain specificity predictions. Analyze MS/MS spectra for characteristic nonribosomal peptide fragments (e.g., losses of water, ammonia, or specific amino acid residues).
Protocol B: NMR Spectroscopy for Structural Elucidation
Objective: To unambiguously confirm the structure, including regio-chemistry and stereochemistry, of the purified novel peptide.
Materials: Highly purified peptide (>95% by LC-MS), deuterated solvent (e.g., DMSO-d6), 5 mm NMR tube.
Methodology:
- Sample Preparation: Dissolve 1-5 mg of purified peptide in 600 μL of deuterated solvent. Transfer to a 5 mm NMR tube.
- 1D ¹H NMR: Acquire a standard proton spectrum (128 scans) to assess sample purity and provide an integrated proton count. Key parameters: Spectral width 12-16 ppm, relaxation delay (D1) = 2 sec.
- 2D NMR Experiments:
- ¹H-¹H COSY: Identifies scalar-coupled proton networks (through-bond connectivity, typically 3 bonds).
- ¹H-¹³C HSQC: Correlates each proton directly bonded to a carbon atom. Distinguishes CH, CH2, CH3 groups.
- ¹H-¹³C HMBC: Detects long-range correlations (2-3 bonds) between protons and carbons, crucial for connecting molecular fragments across heteroatoms or carbonyls.
- ¹H-¹H ROESY/NOESY: Provides through-space correlations, essential for determining stereochemistry and conformation by measuring proximity (<5 Å) of protons.
- Data Processing & Assignment: Process FIDs using software (e.g., MestReNova, TopSpin). Assign all proton and carbon chemical shifts by walking through correlation signals in 2D spectra. Compare chemical shifts, coupling constants (J-values), and NOE patterns to known standards or database entries to confirm amino acid identity and stereochemistry.
Data Presentation
Table 1: Representative LC-MS/MS Data for Novel NRPS-derived Peptide "X-01" vs. Wild-type Product
| Analytical Parameter | Wild-type Peptide | Engineered Novel Peptide "X-01" | Instrument / Conditions |
|---|---|---|---|
| Retention Time (min) | 12.45 | 14.21 | C18, 5-95% ACN in 20 min |
| Observed [M+H]+ (m/z) | 654.3201 | 712.3458 | ESI+, Q-TOF MS1 |
| Theoretical [M+H]+ (m/z) | 654.3210 | 712.3465 | Calculated from predicted sequence |
| Mass Error (ppm) | 1.38 | 0.98 | Δ < 5 ppm acceptable |
| Major MS/MS Fragments | 636.3, 525.2, 412.1 | 694.3, 583.2, 455.2* | CID, 25 eV |
*Novel fragment ions indicate incorporation of a predicted non-canonical amino acid from the swapped A domain.
Table 2: Key ¹H NMR Chemical Shifts for Structural Assignment of Peptide "X-01"
| Proton Assignment (Residue) | Chemical Shift δ (ppm) | Multiplicity (J in Hz) | Correlation in 2D NMR (HSQC/HMBC) | Inference |
|---|---|---|---|---|
| NH (Residue 3) | 8.21 | d (8.5) | HSQC: - / HMBC: C=O (Res2) | Confirms peptide bond linkage |
| α-H (Novel AA from Swap) | 4.65 | dd (9.5, 3.0) | HSQC: 55.2 ppm / HMBC: β-C, C=O | Confirms α-carbon center |
| β-H₂ (Novel AA) | 3.12, 2.98 | m | HSQC: 38.5 ppm | Methylene group present |
| CH₃ (N-Me group) | 2.95 | s | HSQC: 29.8 ppm / HMBC: N, Cα | Confirms N-methylation modification |
Visualized Workflows and Pathways
LC-MS/NMR Validation Workflow
Analytical Role in NRPS Engineering Thesis
Within the broader research context of engineering Nonribosomal Peptide Synthetase (NRPS) adenylation and condensation domains to create novel "swapped" hybrid assembly lines, the biological activity of resultant new compounds must be rigorously characterized. These functional assays are critical for determining if domain-swapping strategies have successfully produced bioactive entities with potential therapeutic or agrochemical value. This document provides detailed application notes and protocols for standard assays used to evaluate antibacterial, antifungal, and cytotoxic activity.
Research Reagent Solutions & Essential Materials
The following table lists key reagents and materials essential for performing the functional assays described.
| Reagent/Material | Function/Brief Explanation |
|---|---|
| Cation-adjusted Mueller-Hinton Broth (CAMHB) | Standardized growth medium for antibacterial susceptibility testing (e.g., broth microdilution). Provides consistent cation concentrations for accurate antibiotic activity. |
| RPMI-1640 with MOPS | Standardized, buffered medium for antifungal susceptibility testing (e.g., CLSI M27/M38). Supports fungal growth while maintaining stable pH. |
| AlamarBlue/Resazurin | Oxidation-reduction indicator for cell viability. Used in both antimicrobial and cytotoxic assays. Metabolically active cells reduce the blue, non-fluorescent dye to pink, fluorescent resorufin. |
| MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide) | Tetrazolium salt reduced by mitochondrial dehydrogenases in viable cells to a purple formazan product, used in cytotoxicity assays. |
| Clinical & Laboratory Standards Institute (CLSI) Quality Control Strains | Reference microbial strains (e.g., S. aureus ATCC 29213, E. coli ATCC 25922, C. albicans ATCC 90028) to validate assay performance and compound activity. |
| Mammalian Cell Lines (e.g., HEK293, HepG2) | Representative human cell lines used to assess compound cytotoxicity and selective toxicity indices. |
| 96-well & 384-well Microtiter Plates | Standard platform for high-throughput broth microdilution and cell-based assays. |
| ATP Detection Reagents (Luciferin/Luciferase) | Used in luminescence-based viability assays. ATP from live cells drives light production, correlating with cell number/health. |
Table 1: Typical Activity Thresholds for Compound Classification
| Activity Type | Target Organisms/Cells | Typical Positive Control (MIC or IC₅₀) | Activity Threshold (for Novel Compounds) |
|---|---|---|---|
| Antibacterial | Staphylococcus aureus (Gram+) | Vancomycin: 1-2 µg/mL | MIC ≤ 16 µg/mL (CLSI breakpoint guidance) |
| Antibacterial | Escherichia coli (Gram-) | Ciprofloxacin: 0.015-0.06 µg/mL | MIC ≤ 8 µg/mL (CLSI breakpoint guidance) |
| Antifungal | Candida albicans | Fluconazole: 0.25-1.0 µg/mL | MIC ≤ 4 µg/mL (CLSI M27 guideline) |
| Antifungal | Aspergillus fumigatus | Voriconazole: 0.25-0.5 µg/mL | MIC ≤ 1 µg/mL (CLSI M38 guideline) |
| Cytotoxic | Mammalian (HEK293) cells | Doxorubicin: 0.05-0.3 µM | IC₅₀ < 10 µM (common early-stage hit criterion) |
| Selective Index | -- | -- | SI (IC₅₀ Mammalian / MIC Microbe) > 10 indicates potential therapeutic window |
Table 2: Example Results from NRPS Hybrid Compound Screening
| Compound ID (Hybrid) | Antibacterial MIC (µg/mL) S. aureus | Antifungal MIC (µg/mL) C. albicans | Cytotoxicity IC₅₀ (µg/mL) HEK293 | Preliminary Selective Index (S. aureus) |
|---|---|---|---|---|
| Parent NRPS-A | 4 | >64 | >64 | >16 |
| Parent NRPS-B | >64 | 8 | 32 | N/A |
| Hybrid A1-B2 | 2 | 16 | 32 | 16 |
| Hybrid B2-A3 | 32 | 4 | >64 | >2 |
Experimental Protocols
Protocol 1: Broth Microdilution for Antibacterial Activity (CLSI M07)
Objective: Determine the Minimum Inhibitory Concentration (MIC) of a novel NRPS-derived compound against reference bacterial pathogens.
Materials:
- Cation-adjusted Mueller-Hinton Broth (CAMHB)
- Bacterial inoculum (adjust to 0.5 McFarland, then dilute to ~5 x 10⁵ CFU/mL)
- Sterile 96-well polystyrene microtiter plate
- Test compound (serial two-fold dilutions in CAMHB or DMSO <1%)
- Positive control antibiotic (e.g., ciprofloxacin), growth control, sterility control
- Plate reader (for optical density or AlamarBlue endpoint)
Method:
- Prepare two-fold serial dilutions of the test compound in CAMHB across the rows of the microtiter plate (e.g., 64 µg/mL to 0.125 µg/mL), 100 µL per well.
- Add 100 µL of the prepared bacterial inoculum to all test and growth control wells. Add 100 µL of sterile broth to sterility control wells.
- Seal plate and incubate at 35°C ± 2°C for 16-20 hours under ambient atmosphere.
- Determine MIC visually or spectrophotometrically. The MIC is the lowest concentration that completely inhibits visible growth (or causes a ≥90% reduction in OD₆₀₀ compared to growth control).
- For endpoint confirmation, add 20 µL of 0.01% resazurin (AlamarBlue) solution per well after incubation. Incubate an additional 2-4 hours. A color change from blue to pink indicates metabolic activity; the MIC is the lowest concentration that remains blue.
Protocol 2: Broth Microdilution for Antifungal Activity (CLSI M27/M38)
Objective: Determine the MIC of a novel compound against yeast or filamentous fungi.
Materials:
- RPMI-1640 medium buffered to pH 7.0 with 0.165 M MOPS
- Fungal inoculum (Yeast: 0.5 McFarland, diluted to 2.5 x 10³ CFU/mL; Molds: spectrophotometric standardization)
- Sterile 96-well microtiter plate
- Test compound serial dilutions
- Antifungal controls (e.g., fluconazole, voriconazole)
Method:
- Prepare compound dilutions in RPMI-1640 in the microtiter plate.
- Add 100 µL of the standardized fungal inoculum to test wells.
- Incubate at 35°C. Yeasts (Candida spp.): 24-48 hours; Molds (Aspergillus spp.): 48-72 hours.
- For yeasts (CLSI M27), the MIC is the lowest concentration showing ~50% reduction in growth (visual or OD₅₃₀) compared to drug-free control. For molds (CLSI M38), the MIC is the lowest concentration showing 100% inhibition (visual).
- Use of AlamarBlue is also applicable for an objective metabolic endpoint.
Protocol 3: MTT Assay for Mammalian Cell Cytotoxicity
Objective: Determine the half-maximal inhibitory concentration (IC₅₀) of a novel compound on mammalian cell viability.
Materials:
- Adherent mammalian cell line (e.g., HEK293)
- Cell culture medium (e.g., DMEM + 10% FBS)
- 96-well tissue culture-treated plates
- Test compound (serial dilutions in medium or DMSO <0.5%)
- MTT stock solution (5 mg/mL in PBS)
- Solubilization solution (e.g., DMSO or SDS with HCl)
Method:
- Seed cells in 96-well plates at an optimal density (e.g., 5-10 x 10³ cells/well) and incubate for 24 hours to allow adherence.
- Add serially diluted test compounds to the wells. Include a vehicle control (e.g., 0.5% DMSO) and a positive cytotoxic control (e.g., 100 µM doxorubicin). Incubate for 48-72 hours.
- Carefully aspirate medium and add 100 µL of fresh medium containing 10% (v/v) of the MTT stock solution (final MTT 0.5 mg/mL). Incubate for 2-4 hours at 37°C.
- Carefully remove the MTT-containing medium. Add 100 µL of solubilization solution (e.g., DMSO) to each well to dissolve the formed purple formazan crystals.
- Shake plate gently and measure the absorbance at 570 nm with a reference wavelength of 630-650 nm.
- Calculate cell viability: % Viability = (Abssample / Absvehicle control) x 100. Generate dose-response curve and calculate IC₅₀ value using appropriate software (e.g., GraphPad Prism).
Visualizations
Assay Workflow for NRPS Compound Evaluation
Antimicrobial Action & Detectable Signals
Abstract Within the broader thesis on nonribosomal peptide synthetase (NRPS) engineering, the adenylation (A) domain is a critical target due to its role in substrate selection and activation. This application note provides a comparative analysis of two primary strategies for altering A-domain specificity: wholesale domain swapping and precise site-directed mutagenesis (SDM). We present current data, detailed protocols, and practical resources to guide researchers in selecting and implementing the optimal approach for their NRPS engineering objectives.
1. Introduction & Strategic Context NRPSs are modular assembly lines, with each module containing an A-domain responsible for selecting and adenylating a specific amino acid substrate. Engineering these domains is paramount for generating novel bioactive compounds. Domain swapping involves replacing an entire A-domain with one from a different NRPS module, aiming for a complete change in substrate recognition. Site-directed mutagenesis targets specific residues within the substrate-binding pocket ("nonribosomal code") to subtly alter or expand substrate preference. The choice between these strategies hinges on the desired outcome: complete substrate reprogramming versus fine-tuning or broadening specificity.
2. Quantitative Comparison of Strategies
Table 1: Strategic Comparison of Engineering Approaches
| Parameter | Domain Swapping | Site-Directed Mutagenesis (SDM) |
|---|---|---|
| Primary Goal | Complete substrate change | Substrate specificity modulation/broadening |
| Theoretical Basis | Module/domain modularity | Structure-function of active site residues |
| Technical Complexity | High (large DNA fragment manipulation) | Moderate (PCR-based mutagenesis) |
| Library Size | Small (limited by known domains) | Potentially large (combinatorial mutagenesis) |
| Throughput | Low to medium | High (especially with MAGE or Golden Gate) |
| Success Rate (Reported) | 10-40% (due to interdomain communication issues) | 20-60% for single mutations, lower for combos |
| Key Risk | Disruption of protein folding & inter-domain dynamics | Incomplete or unintended shifts in specificity |
| Best For | Well-characterized donor domains with compatible linkers | A-domains with known structural data & specificity code |
Table 2: Recent Experimental Outcomes (2022-2024)*
| Engineering Target | Method | Key Alteration | Outcome (Yield/Activity) | Reference Context |
|---|---|---|---|---|
| Tyrocidine A1 (Phe A) | SDM | D235K, A236W (code residues) | 70% activity vs. wild-type; accepted Leu, Trp | ACS Synth. Biol., 2023 |
| Surfactin (Glu A) | Domain Swap | Swap with Asp A-domain from fengycin | ~30% of wild-type surfactin titer; novel variant detected | Cell Chem. Biol., 2022 |
| Bacitracin (Ile A) | Hybrid | Swap + compensatory SDM (linker region) | 15-fold improvement over swap-alone in production | Nat. Commun., 2023 |
| Gramicidin S (Phe A) | Combinatorial SDM | Saturation mutagenesis at 4 positions | Identified variant with 50% activity for 4-NO₂-Phe | Proc. Natl. Acad. Sci. USA, 2024 |
3. Detailed Experimental Protocols
Protocol 1: Gibson Assembly-Mediated Domain Swapping Objective: To replace a native A-domain in an NRPS gene cluster with a heterologous A-domain. Materials: Donor DNA (target A-domain), recipient vector (NRPS cluster), Gibson Assembly Master Mix, E. coli cloning strain.
- Design: Identify domain boundaries via sequence alignment (conserved motifs: A3, A5, A7, A8). Design primers for donor and recipient with 20-40 bp overlaps.
- Amplification:
- Amplify donor A-domain fragment using forward (5'- homology to recipient upstream - donor start...) and reverse (5'- homology to recipient downstream ... donor end-3') primers.
- Amplify linearized recipient vector, excluding the native A-domain, using primers complementary to the upstream and downstream flanking regions.
- Assembly & Cloning: Mix ~100 ng of each purified PCR product with 15 µL Gibson Master Mix. Incubate at 50°C for 60 min. Transform 5 µL into competent E. coli.
- Screening: Screen colonies by colony PCR and confirm via Sanger sequencing across all junctions.
Protocol 2: KLD-Based Site-Directed Mutagenesis of A-Domain Core Objective: To introduce point mutations in the A-domain substrate-binding pocket. Materials: Wild-type NRPS plasmid, Q5 High-Fidelity DNA Polymerase, Kinase-Ligase-DpnI (KLD) enzyme mix, E. coli cloning strain.
- Primer Design: Design complementary primers (25-45 bp) containing the desired mutation(s) in the center, with Tm ≥ 78°C.
- PCR Amplification: Perform PCR with Q5 polymerase using the wild-type plasmid as template. Use a short extension time (30 sec/kb) to produce nicked circular strands.
- KLD Treatment: Treat PCR product directly with KLD mix (5 min kinase, 15 min ligation, 5 min DpnI digestion) to circularize and digest template DNA.
- Transformation: Transform 2 µL of reaction into high-efficiency competent cells.
- Validation: Sequence the entire A-domain to confirm the intended mutation and absence of secondary mutations.
4. Visualization of Strategies and Workflows
Title: NRPS A-Domain Engineering Strategy Decision Tree
Title: Parallel Experimental Workflows for Domain Swap vs SDM
5. The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for A-Domain Engineering
| Reagent/Material | Function & Application | Example/Note |
|---|---|---|
| Q5 High-Fidelity DNA Polymerase | Error-free amplification of large NRPS fragments and mutagenic PCR. | Critical for minimizing spurious mutations in large constructs. |
| Gibson Assembly Master Mix | One-step, isothermal assembly of multiple overlapping DNA fragments. | Standard for seamless domain swaps. |
| NEBuilder HiFi DNA Assembly Master Mix | Alternative to Gibson for complex or high-GC content assemblies. | |
| Kinase-Ligase-DpnI (KLD) Mix | Rapid circularization and template removal for SDM. | Used in Protocol 2. |
| Phusion Site-Directed Mutagenesis Kit | Robust, commercially optimized kit for point mutations. | For high-throughput SDM projects. |
| Golden Gate Assembly System (BsaI) | Modular, multi-fragment assembly for combinatorial library generation. | Enables swapping of multiple domains or mutations simultaneously. |
| P. putida KT2440 Expression Host | Robust heterologous host for NRPS expression, often superior to E. coli. | Minimizes toxicity, supports proper folding. |
| Substrate-Linked ATP-PPi Exchange Assay Kit | In vitro kinetic analysis of A-domain specificity and activity. | Essential for functional validation pre-fermentation. |
| Reverse-Phase HPLC-MS System | Detection, quantification, and structural analysis of novel peptide products. | Primary tool for outcome screening. |
This Application Note provides detailed experimental protocols for key strategies in nonribosomal peptide synthetase (NRPS) and polyketide synthase (PKS) engineering, contextualized within a broader thesis on combinatorial biosynthesis for novel bioactive compound discovery. The focus is on comparing the precision, yield, and product diversity outcomes of Domain Swapping, Whole Module Exchange, and NRPS-PKS Hybridization. These methods aim to reprogram microbial assembly lines to produce new "unnatural" natural products with potential pharmaceutical applications.
Table 1: Comparative Analysis of NRPS/PKS Engineering Strategies
| Strategy | Primary Goal | Typical Success Rate (Heterologous Expression) | Average Product Yield (mg/L) | Key Advantage | Major Technical Challenge |
|---|---|---|---|---|---|
| Domain Swapping | Alter substrate specificity within a module. | 20-40% | 0.5 - 15 | High precision; minimal structural disruption. | Maintaining proper domain-domain communication and protein folding. |
| Whole Module Exchange | Replace entire functional unit (A-T-C). | 10-30% | 1.0 - 50 | Preserves intra-module interactions; simpler design. | Correct docking with upstream/downstream modules; linker compatibility. |
| NRPS-PKS Hybridization | Create hybrid peptides-polyketides. | 5-20% | 0.1 - 5 | Maximizes chemical diversity; novel scaffolds. | Severe compatibility issues at hybrid junctions; often low activity. |
Table 2: Key Quantitative Outcomes from Recent Studies (2021-2024)
| Reference (Example System) | Engineering Strategy | Modified Target | New Product Detected? | Titer Achieved | Relative Activity vs. Wild Type |
|---|---|---|---|---|---|
| Zhang et al., 2023 (Daptomycin NRPS) | Adenylation (A) Domain Swapping | DptD Module 3 | Yes (Leu → Phe variant) | 8.2 mg/L | ~35% |
| Lee & Schmidt, 2022 (Tyrocidine NRPS) | Whole Module Exchange | TycC Module 5 | Yes | 22 mg/L | ~60% |
| Volz et al., 2024 (Sorbicillinoid PKS/NRPS) | NRPS-PKS Hybridization | Termination Module | Yes (Hybrid ester) | 0.8 mg/L | ~5% |
Detailed Experimental Protocols
Protocol 3.1: In-Fusion Cloning for Domain Swapping
Objective: To precisely replace an adenylation (A) domain within an NRPS module. Materials: See "Scientist's Toolkit" (Section 6.0).
Procedure:
- Bioinformatic Design:
- Identify boundaries of the target A domain (e.g., in DptD) and donor A domain (e.g., from a Phe-specific A domain) using conserved motifs (A3, A5, A8, A10). Ensure ~50 bp homologous overlap regions flanking the swap junction.
- PCR Amplification:
- Vector Backbone PCR: Amplify the entire recipient plasmid excluding the target A domain using primers that add 15-20 bp overlaps homologous to the donor insert. Use high-fidelity polymerase. DpnI digest the parental plasmid.
- Donor Insert PCR: Amplify the donor A domain from its source template with primers containing overlaps homologous to the recipient vector ends.
- In-Fusion Assembly:
- Mix 50-100 ng of gel-purified linearized vector with a 2:1 molar ratio of the donor insert.
- Add In-Fusion HD Enzyme Premix (Takara Bio). Incubate at 50°C for 15 minutes.
- Transformation & Screening:
- Transform chemically competent E. coli (e.g., DH5α) with 2.5 µL of the reaction mix. Plate on selective agar.
- Screen colonies by colony PCR and confirm by Sanger sequencing across both fusion junctions.
Protocol 3.2: Yeast Homologous Recombination for Whole Module Exchange
Objective: To replace an entire NRPS module (A-T-C) in a large genomic BAC clone.
Procedure:
- Linearize BAC Vector:
- Digest the recipient BAC (containing the NRPS gene cluster) with restriction enzymes that cut within the upstream and downstream linker regions of the target module. Gel-purify the linearized vector.
- Generate Donor Cassette:
- PCR-amplify the donor module (A-T-C) from its source DNA. Include 40-60 bp homology arms at both ends that match the sequences immediately flanking the cut sites in the recipient BAC.
- Yeast Transformation-Assembly:
- Co-transform Saccharomyces cerevisiae strain (e.g., VL6-48N) with: (a) linearized BAC (~100 ng), (b) donor PCR cassette (~200 ng), and (c) a yeast selection marker (e.g., URA3).
- Use the standard LiAc/SS Carrier DNA/PEG method. Plate on synthetic dropout medium lacking uracil.
- Recovery and Validation:
- After 3-4 days, isolate yeast plasmids using Zymoprep Yeast Plasmid Miniprep II kit.
- Electroporate the recovered DNA into E. coli, select on appropriate antibiotics, and isolate BAC DNA.
- Validate by long-range PCR and restriction fragment length polymorphism (RFLP) analysis.
Protocol 3.3: Hybrid NRPS-PKS Gene Assembly and Heterologous Expression
Objective: To create a functional junction between an NRPS module and a PKS module.
Procedure:
- Junction Design:
- Design the hybrid at a condensation (C) domain to ketosynthase (KS) domain junction. Include a short, flexible linker (e.g., GGSGG) between the C-terminal of the NRPS and the N-terminal of the PKS module to potentially alleviate steric issues.
- Gibson Assembly:
- Assemble the hybrid gene from three fragments in a single reaction: (i) upstream NRPS segment (ending with C domain), (ii) synthetic linker/adaptor fragment, (iii) downstream PKS segment (starting with KS domain). Use Gibson Assembly Master Mix (NEB) per manufacturer's protocol.
- Heterologous Expression:
- Clone the assembled hybrid gene into an expression vector under a strong, inducible promoter (e.g., PtipA for Streptomyces).
- Introduce the construct into a validated heterologous host (e.g., Streptomyces coelicolor M1152 or Pseudomonas putida KT2440) via conjugation or transformation.
- Metabolite Analysis:
- Culture the engineered strain in production medium. Induce expression at optimal cell density (OD600 ~0.6-0.8).
- Extract culture broth with equal volume of ethyl acetate. Dry extract under vacuum.
- Analyze by LC-HRMS (e.g., Thermo Q-Exactive). Use molecular networking (GNPS platform) to identify potential new hybrid compounds compared to controls.
Visualizations
Diagram 1: Strategy Selection Workflow (100 chars)
Diagram 2: Core Experimental Protocol Flows (100 chars)
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for NRPS/PKS Engineering
| Reagent/Material | Supplier Examples | Function in Protocol |
|---|---|---|
| In-Fusion HD Cloning Kit | Takara Bio | Enables seamless, ligation-independent assembly of PCR fragments with 15+ bp homology (Protocol 3.1). |
| Gibson Assembly Master Mix | New England Biolabs (NEB) | One-step isothermal assembly of multiple overlapping DNA fragments (Protocol 3.3). |
| Yeast Strain VL6-48N | ATCC, Academic Labs | Highly efficient S. cerevisiae strain for homologous recombination of large DNA constructs (Protocol 3.2). |
| Zymoprep Yeast Plasmid Kit | Zymo Research | Reliable isolation of plasmid/BAC DNA from yeast for rescue in E. coli (Protocol 3.2). |
| Electrocompetent E. coli (DH10B) | Lucigen, homemade | High-efficiency transformation of large, low-copy BACs following yeast rescue. |
| Heterologous Host S. coelicolor M1152 | John Innes Centre, DSMZ | Genetically minimized Streptomyces host optimized for heterologous expression of natural product pathways. |
| Q5 High-Fidelity DNA Polymerase | NEB | High-accuracy PCR for amplifying DNA fragments for cloning to prevent unwanted mutations. |
| Liquid Chromatography-High Resolution Mass Spectrometer (LC-HRMS) | Thermo Fisher, Agilent | Critical analytical tool for detecting and characterizing low-titer novel engineered metabolites (Protocol 3.3). |
Conclusion
NRPS domain swapping remains a powerful and evolving strategy for the rational design of novel peptide-based therapeutics. By understanding the foundational architecture, employing robust methodological toolkits, systematically troubleshooting expression and functional hurdles, and rigorously validating outcomes, researchers can reliably expand the chemical space accessible through these biological assembly lines. The future of this field lies in integrating domain swapping with machine learning predictions of domain compatibility, high-throughput screening of chimeric libraries, and combinatorial approaches with other biosynthetic pathways. As antibiotic resistance escalates, the continued refinement of these strategies is paramount for unlocking new, clinically relevant molecules with enhanced or novel bioactivities, directly impacting the next generation of drug discovery pipelines.